California Jumps Into the Drug Business
The bio-pharmaceutical industry, including the biotechnology sector, is in for another shock.
Taking a page off Elizabeth Warren’s book, California Governor Gavin Newson announced his plan to create the first-ever state-run prescription drug label in the country.
This move would be an ideal way to leverage the size and power of the California population in terms of negotiating advantageous pricing with generic drug manufacturers.
Wielding California’s massive purchasing power courtesy of the 40 million residents, almost 33% of which are enrolled in Medicaid or Medicare, Newsom believes that the government can get generic drugs for substantially lower prices from the manufacturers. This idea is part of Newsom’s broader vision to lower the healthcare costs in California.
In effect, the Golden State is stepping in where the federal government has failed to act. Washington should have used their massive purchasing power as a cudgel to lower prices decades ago. We’ve seen a half-century of talk, but no action.
Research from the Kaiser Family Foundation shows that 6 out of 10 Americans take a prescription, with 79% of them complaining about the unreasonable costs of these drugs.
As a result of these prohibitive prices, 3 in 10 Americans no longer take their prescribed medications adding hugely to the nation’s health care bill.
Even governments find it challenging to keep up with the costs of healthcare, with California’s Medicaid program for the less fortunate, otherwise known as Medi-Cal, reached over $100 billion annually in state and federal spending alone.
Inevitably, the skyrocketing prices pushed people to look at generic medications as more reasonable alternatives to brand name medications.
However, Newsom’s announcement wasn’t met with an overwhelmingly positive response from the generics sector either.
In fact, the Association for Accessible Medicines, the advocacy organization for generic drug manufacturers, only sent out a polite message regarding Newson’s announcement.
The group said that they "look forward to working with California to help expand access to safe, affordable and effective generic and biosimilar medicines, but let's make our decisions and policies based on the best data and science available." You couldn’t get any more anodyne.
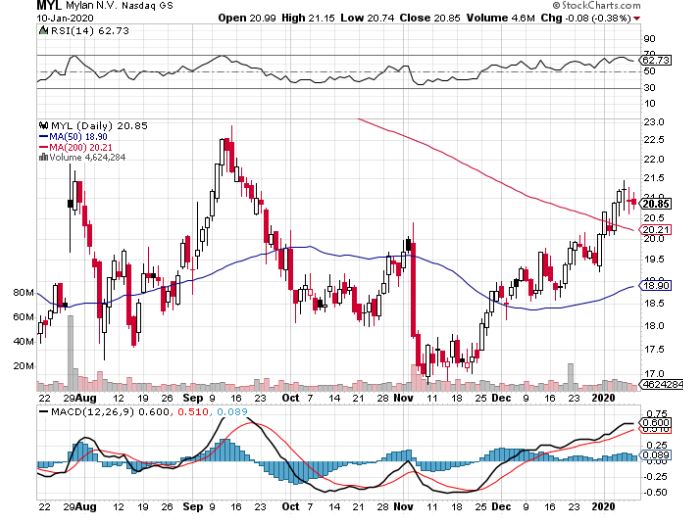
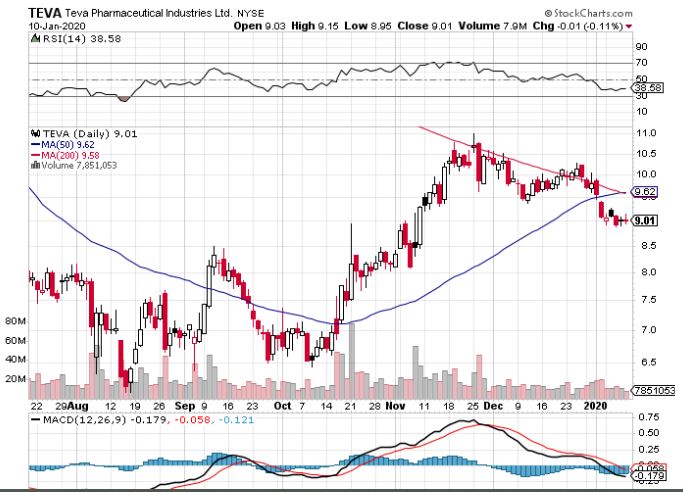
Despite their lukewarm response to this plan, two major players in the generic pharmaceutical industry have already been identified as the front runners for this shift to generics movement: Mylan (MYL) and Teva Pharmaceutical Industries (TEVA).
To say that 2019 was an awful one for Mylan is an understatement. The generic drugmaker faced a slew of issues including declining sales in their North American market, endless legal battles, and even exclusivity loss for its top-selling impotence treatment Tadalafil (Cialis).
However, Mylan moved to turn things around in July 2019 when it entered a merger agreement with Pfizer’s (PFE) generic unit Upjohn. The two companies will turn into one entity, called Viatris, and will be launched by mid-2020.
According to the terms of this deal, Viatris will be handling the off-patent but lucrative branded drugs of Pfizer such as cholesterol treatment Lipitor, pain medication Celebrex, and erectile dysfunction and the blue erectile dysfunction drug Viagra.
This agreement aims to inject more money into Mylan’s research and development team for them to create more complex generics and biosimilars.
At the same time, the new company will already have a “ready-made” tried and tested drug portfolio courtesy of Pfizer’s off-patent previous blockbusters.
Apart from the Pfizer’s lineup, Mylan also has a number of key products to contribute to Viatris.
One is a biosimilar of Roche’s (RHHBY) blockbuster breast cancer drug Herceptin (it added four years to the life of my first wife). The launches of promising products, like biosimilar versions of Teva’s multiple sclerosis injection Copaxone and GlaxoSmithKline’s (GSK) asthma medication Advair, are also positive indicators of growth.
With these decisions, Mylan is expected to have a brighter 2020.
Another generic drug maker that’s expected to make a comeback in 2020 is Teva.
In 2019, everything that could possibly go wrong went wrong for this company. As a refresher, here’s what it had to endure: executive leadership turnover, bribery allegations, generic competition for its top-selling drug Copaxone, and of course, the opioid lawsuits from 44 states.
Needless to say, Teva’s profit estimates tumbled and its dividend was shelved. Worst, its debt load has been a huge warning sign that repelled investors left and right.
However, Teva has managed to turn things around.
The opioid lawsuits are reaching a reasonable settlement and the company now has a stronger leadership team. More importantly, it has been successful in cutting down its expenditures. If things go smoothly, Teva is expected to save $3 billion in yearly expenses to improve its profit margins in 2020.
On top of these, Teva has regained its footing in the generic drug market via the steady climb in terms of sales of its newer branded treatments, Ajovy and Austedo.
All in all, the stage is set for Teva to make its comeback and patient investors should expect to be richer in 2020.
Since both generic drug makers churned out less than stellar numbers in 2019, the stocks are likely undervalued at the moment. Hence, it’s important to take advantage of this and buy before these regain momentum and the prices skyrocket once again.