The Future of Regenerative Medicine
As our bodies begin to show signs of aging and fatigue, exploring ways to regenerate our organs has become crucial in ensuring a hale and hearty lifespan.
This demand has given rise to a branch of biotechnology and healthcare, which could very well be on the brink of becoming the next big thing in the mainstream biopharmaceutical industry: regenerative medicine.
For example, experts at Gladstone Institutes is applying gene therapy to repair heart damage. Basically, their goal is to reprogram scar tissue and transform it into a new heart cell.
What they do is inject the genes into the damaged heart caused by a heart attack. Then, these “new” genes alter the scar cells, converting them into beating hearts.
This approach no longer demands any cloning to obtain an extra set of organs. The Gladstone Institutes’ use of gene therapy allows us to regrow our own set of organs right inside our bodies.
And in case you’re wondering whether this type of work actually has a future or just another trend that would quietly disappear in the future, I’m telling you that this industry has incredibly potential.
Just look at the $12 billion valuation of a biotechnology unicorn called Samumed in San Diego.
Founded in 2008, this company has spent most of its lifetime operating under the radar. It impressively came out of the shadows last 2016 and was quickly dubbed as an “anti-aging” company.
To them, though, they’re a “de-aging” company. That is, they believe that people should be brought back to their peak health conditions before they can even begin to restore youth.
This ideology is exhibited by the company’s lead program: a knee osteoarthritis cure called Lorecivivint.
As we know, osteoarthritis has no known treatment that works to reverse the damage to the joint.
That’s why the doctors focus on handling or managing the symptoms. They tell their patients to exercise and lose weight to boost muscle strength and decrease the burden on their joints.
They also prescribe various drugs like painkillers, some anti-inflammatory pills, and even cortisone shots. Other patients would eventually need to go through replacement surgery.
This is where Samumed comes in.
The company created Lorecivivint to repair the joint damage. That way, patients will no longer need to go through all the burden of managing the symptoms of knee osteoarthritis.
In their proof-of-concept report, Samumed shared that one year after getting injected with Lorecivivint, the X-rays of the knees of the patients showed that there was an increase in “medial compartment joint space width.”
In simpler terms, the knees grew cartilage after a single shot of Lorecivivint.
Other than working on a cure for knee osteoarthritis, Samumed is also looking into treating male pattern baldness.
Another impressive biotechnology company focused on regenerative medicine is Humacyte, which recently shifted from being a clinical-stage firm to a commercial one.
Humacyte’s core work is on Human Acellular Vessels (HAVs) or “implantable regenerative human tissue.”
A use case for this is when a patient has damaged blood vessels. Typically, there are three options to treat this: take a vessel from another part of the body, try to implant a donated vessel, or utilize a plastic tube.
The first one requires at least two surgeries and, of course, losing a vessel in another part of your body.
Meanwhile, the second and third options expose the patient to the possibility of an infection or the body rejecting the vessel or plastic tube.
Humacyte’s HAVs offer a fourth option.
Since the HAVs carry similar properties as the native tissues of the patient’s body, they significantly lower the risk of rejection.
Basically, they’re “growing” HAVs that won’t be rejected by the body.
More importantly, the company is creating engineered off-the-shelf replacement tissue that can be implanted to anyone without using immunosuppressive drugs.
This is impressive because immunosuppressants are staples in ensuring that the body does not reject the organs. However, the use of this can be dangerous because it increases the risk of infections.
So far, Humacyte has been working on coming up with safer and more effective treatments for hemodialysis patients since the current methods tend to expose them to higher risks of infections.
If everything goes according to plan, then the company will be able to file for FDA approval by 2022.
While the technologies offered in the regenerative medicine space have been discussed and even praised for years, it’s only recently that these became commercially viable.
For all the noise and hype surrounding these breakthrough and next-generation treatments, only a handful of patients have actually benefited from them.
However, 2021 might just mark the year that all these will change.
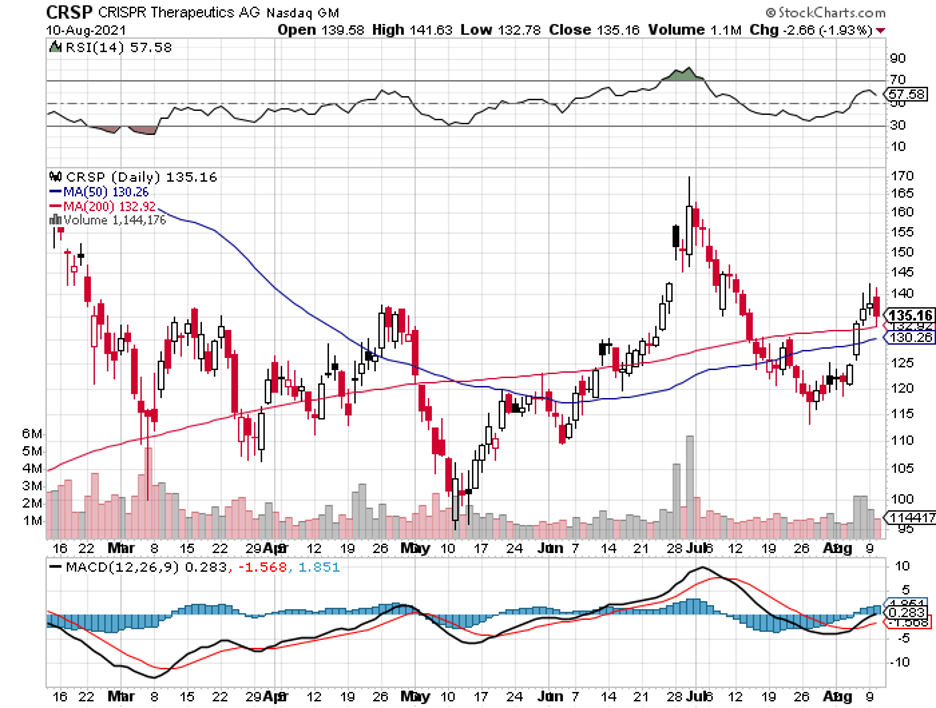
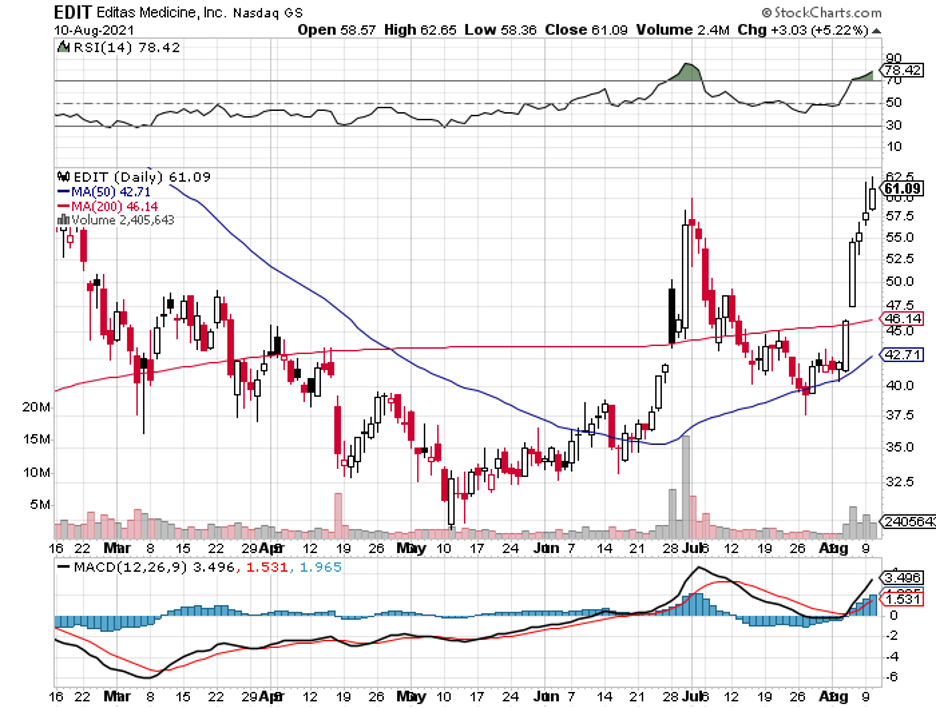
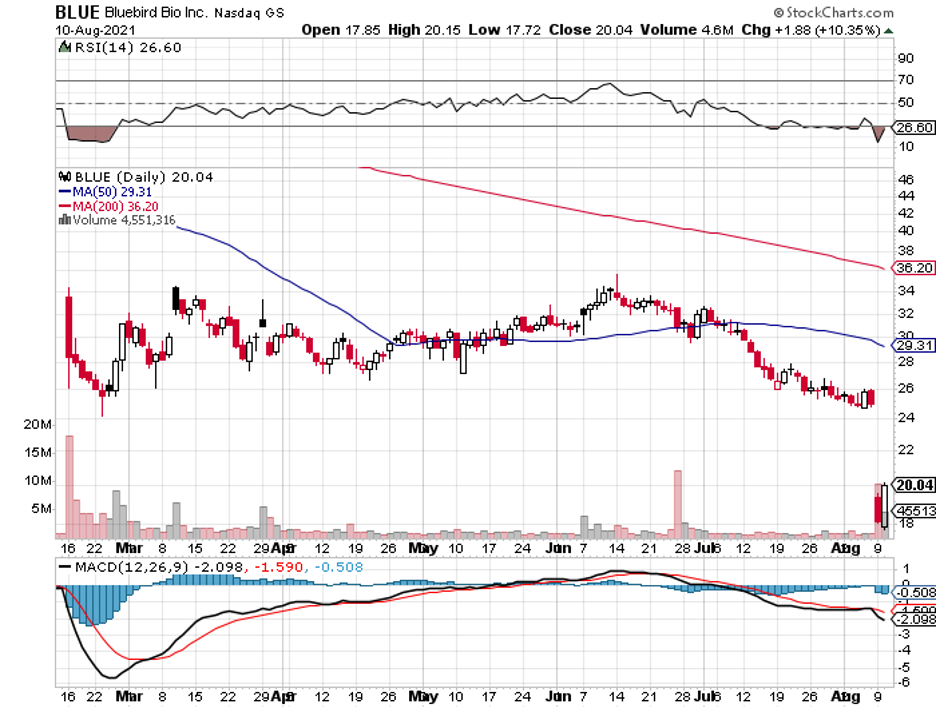
Other than the private firms and smaller biotechnology companies like CRISPR Therapeutics (CRSP), Editas Medicine (EDIT), and bluebird Bio (BLUE), bigger names in the biopharmaceutical space, including Pfizer (PFE), AstraZeneca (AZN), GlaxoSmithKline (GSK), Takeda Pharmaceuticals (TAK), and Regeneron (REGN), are also starting to invest more aggressively into it.