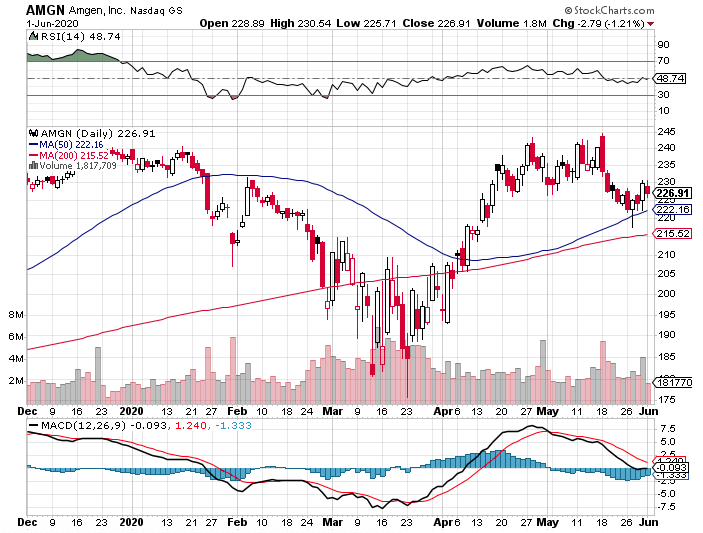
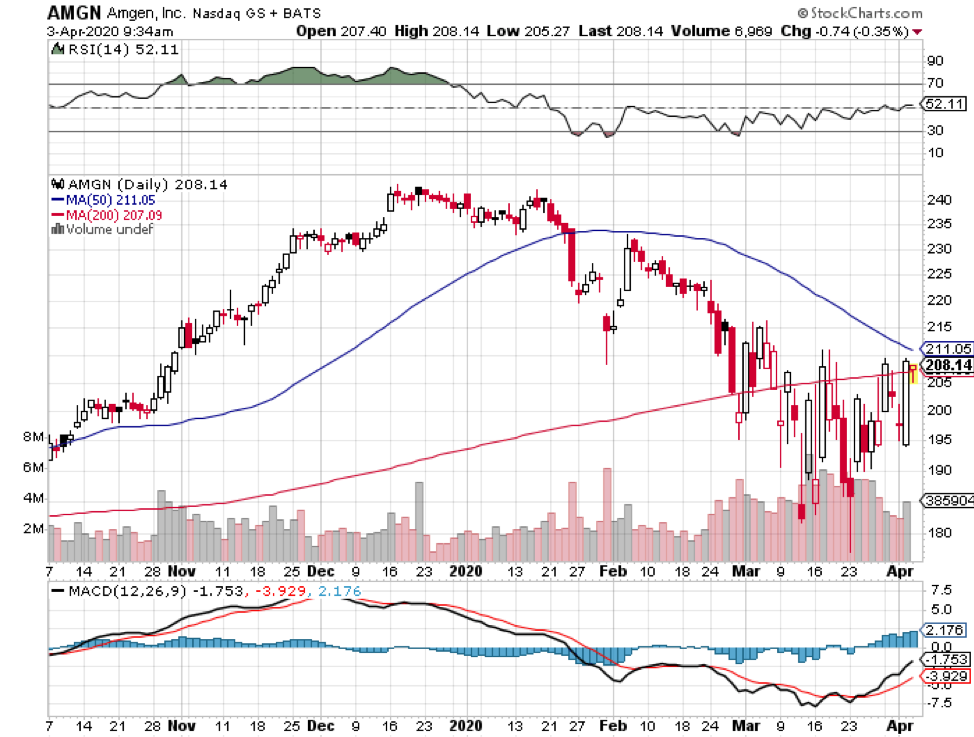
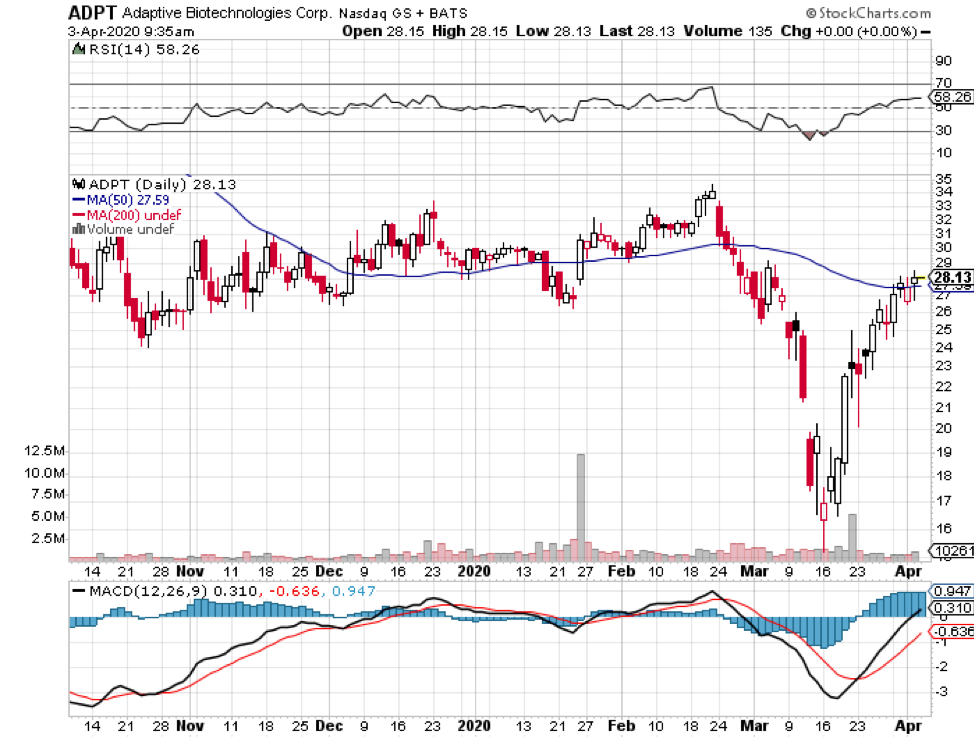
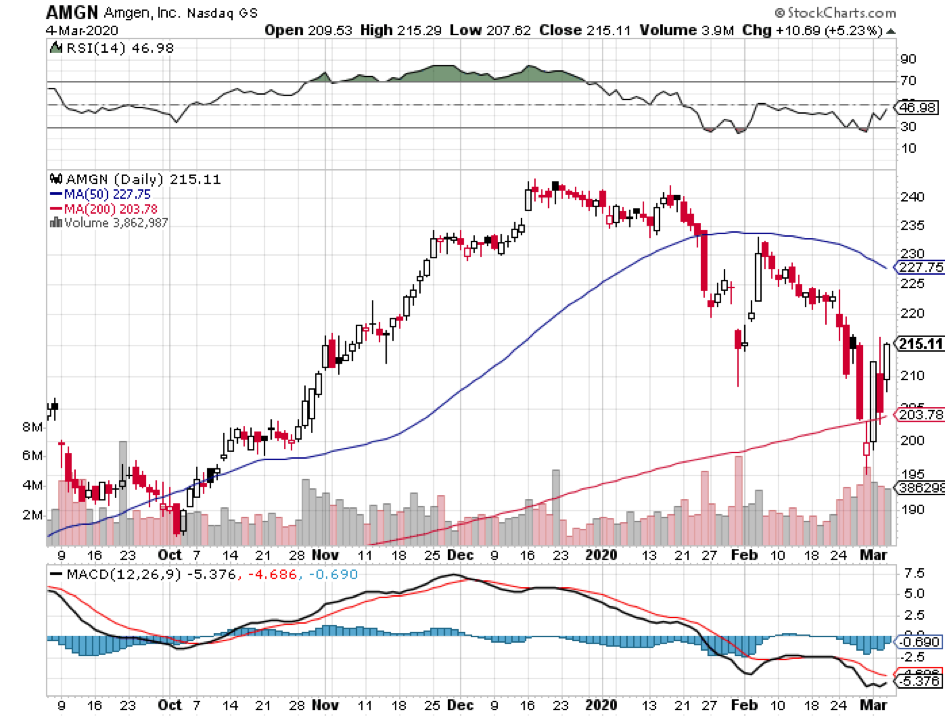
In this current coronavirus market, discovering a stock that can survive the pandemic without suffering a major hit is akin to finding the Holy Grail.
This is why I’m on the lookout for undervalued names that offer strong dividends and a stable balance sheet.
In my search, I once again came across the biotechnology pioneer Amgen (AMGN), a 40-year-old company that has continuously proven its critics wrong.
After four decades in the business, this healthcare heavyweight has yet to show any signs of slowing down. If anything, Amgen has been consistent in its efforts to show off its promising pipeline.
In 2020 alone, the company reported at least 39 drug candidates in its pipeline, with practically half already in Phase 3 trials.
As one of the founding fathers of the biotechnology sector, Amgen, which was founded by my UCLA college biochemistry professor, would be remiss to skip on the coronavirus race. If I’d only stuck with him a little longer, I would be filthy rich by now.
In April, the company announced its collaboration with Adaptive Biotechnologies (ADPT) to develop antibodies to be used for COVID-19 treatment.
While the biotechnology giant isn’t the first to jump into the fray, it has an undeniable ace up its sleeve: Amgen is known as one of the leaders in the development of antibody-based treatments. This alone makes it a strong contender.
Outside its coronavirus work, Amgen has an extensive list of prospects in its pipeline along with a number of workhorse drugs.
However, not everything is as smooth sailing as investors would hope.
Previous blockbusters, rheumatoid arthritis drug Enbrel and chemotherapy medication Neulasta, which comprised 40% of Amgen’s product sales in the first quarter of 2019, failed to move the needle during the same period in 2020. Actually, Neulasta suffered a devastating 40% drop in sales.
Other top performers like multiple myeloma treatment Xgeva and anemia drug Aranesp are having trouble as well, eking out a measly 2% in sales gains in the said period.
As for anemia injection Epogen, the bestselling drug dropped by 29%. Even the sales of hyperparathyroidism medication Cinacalcet slid by 42%.
Overall, this doesn’t sound like an auspicious beginning for Amgen this 2020.
Nonetheless, the company’s robust growth in other areas made up for the laggards.
In fact, Amgen’s total product sales increased by 12%, jumping from $5.3 billion in the first quarter of 2019 to almost $5.9 billion in the same period in 2020.
For instance, sales of osteoporosis treatment Prolia jumped by 10%, pushing the drug in the second spot among the top 10 products in Amgen’s portfolio.
Meanwhile, cholesterol drug Repatha continues to impress, showing off a 62% increase in revenue from $141 million to $229 million.
Even the newer multiple myeloma treatment Kryprolis went up by 19%, while metastatic colorectal cancer injection Vectibix registered a 19% sales gain. Sales for cancer medication Blincyto also went up by 36% year over year to hit $94 million.
Another drug surging forward is kidney treatment Parsabiv, which reported a 39% increase in sales. Newcomer postmenopausal osteoporosis drug Evenity, which was launched in the US market just last year, recorded a respectable $100 million in sales.
In terms of developments that actually pushed the needle for Amgen, the first thing that comes to mind is definitely the acquisition of psoriasis and psoriatic arthritis medication Otezla.
This blockbuster drug, which the company bought for $13.4 billion in cash from the recently acquired Celgene Corporation (CELGN), raked in $479 million in revenues for the first quarter of 2020 alone.
Amgen executives estimate a low double-digit year-over-year sales increase for Otezla up until 2024.
Another exciting development for Amgen is its deal with BeiGene (BGNE), in which the biotechnology pioneer acquired a 20.5% stake in the Chinese company.
Part of this deal is BeiGene’s efforts to commercialize select drugs from Amgen’s oncology lineup to target the expansive Chinese market.
On top of this, the two companies are slated to develop 20 new cancer drugs as well. So far, AMG 510 and tezepelumab are eyed to be launched by 2021.
Amgen is also gearing up to ride the wave of biosimilars, which has a market estimated to surpass $69 billion in the next five years. Due to its lower costs, biosimilars are projected to save consumers roughly $160 billion during the same period.
Perhaps this lucrative opportunity is what made Amgen realize that if they can’t beat them, they might as well join them. That is, the company has already worked towards becoming a major player in the biosimilar space.
In fact, Amgen has started with this plan in 2017 courtesy of the company’s first-ever approved cancer-fighting biosimilar: Mvasi.
So far, Amgen has 10 biosimilars in its current pipeline. Four of which already received FDA approval.
Looking at Amgen’s financial records, it’s safe to say that the company has a strong financial position.
It offers a yield of approximately 2.8% and a payout ratio of over 47%. The dividend coverage ratio is roughly 212%, ensuring a safe dividend and a 5-year growth rate of almost 19%.
Amgen’s profit margin is recorded to be at least 23% in 9 of the recent 10 fiscal years, with growth expected since 1 in 5 cancer patients in the United States uses Amgen medication.
The company’s free cash flow in the first quarter of 2020 rose to $2 billion from the $1.7 billion reported during the same period in 2019.
These quarterly results along with its multitude of growth prospects are proof to Amgen’s capacity to navigate through the decline in other product offerings. The biotechnology company’s numbers in the first quarter of this year demonstrate there’s a good chance it will break through resistance because of its solid sales momentum.
This makes Amgen attractive to long-term biotechnology and healthcare investors. With its strong record, promising pipeline, and decent dividend, the company will be able to sustain its status as a good buy.