Mad Hedge Biotech and Healthcare Letter
February 23, 2023
Fiat Lux
Featured Trade:
(BATTLE FOR GENE THERAPY SUPREMACY)
(CRSP), (NVS), (BIIB), (BLUE), (VYGR), (GBIO), (SIOX), (NTLA), (EDIT), (VRTX), (PRIME), (BEAM)

Mad Hedge Biotech and Healthcare Letter
February 23, 2023
Fiat Lux
Featured Trade:
(BATTLE FOR GENE THERAPY SUPREMACY)
(CRSP), (NVS), (BIIB), (BLUE), (VYGR), (GBIO), (SIOX), (NTLA), (EDIT), (VRTX), (PRIME), (BEAM)

Gene therapy is arguably one of the most fascinating and revolutionary fields in the healthcare and biotechnology industry.
A significant reason for the excitement behind gene therapy is that it provides the possibility of “functional cures,” such as “one-and-done treatments,” for patients. It’s also why these therapies are some of the most costly on the market.
For example, Zolgensma from Novartis (NVS), which focuses on treating spinal muscular atrophy in infants, has a whopping $2 million-plus price tag. Despite that, it’s considered the best option.
For context, its counterpart, Spinraza from Biogen (BIIB), costs roughly $750,000 in the first year of treatment. Unlike Zolgensma, Spinraza needs to be administered four times each year. After the first treatment, patients would need to pay $350,000 per annum. By the fifth year, Spinraza has surpassed the treatment cost of Zolgensma.
Despite its incredible potential, gene therapy is one of the riskiest bets.
Take Bluebird Bio (BLUE) into consideration. This biotech has won not only one but two regulatory approvals for its innovative gene therapies. One is for Skysona, which targets a rare cerebral condition called adrenoleukodystrophy; the other, Zynteglo, is for the blood disorder beta-thalassemia. Unfortunately, this biotech’s price has slid by more than 90% in the past five years.
Working on gene therapies is filled with complicated and challenging obstacles. Most companies in this segment ended up burning through their cash without successfully launching a marketable product. Some examples of these are Voyager Therapeutics (VYGR), Generation Bio (GBIO), and Sio Gene Therapies (SIOX).
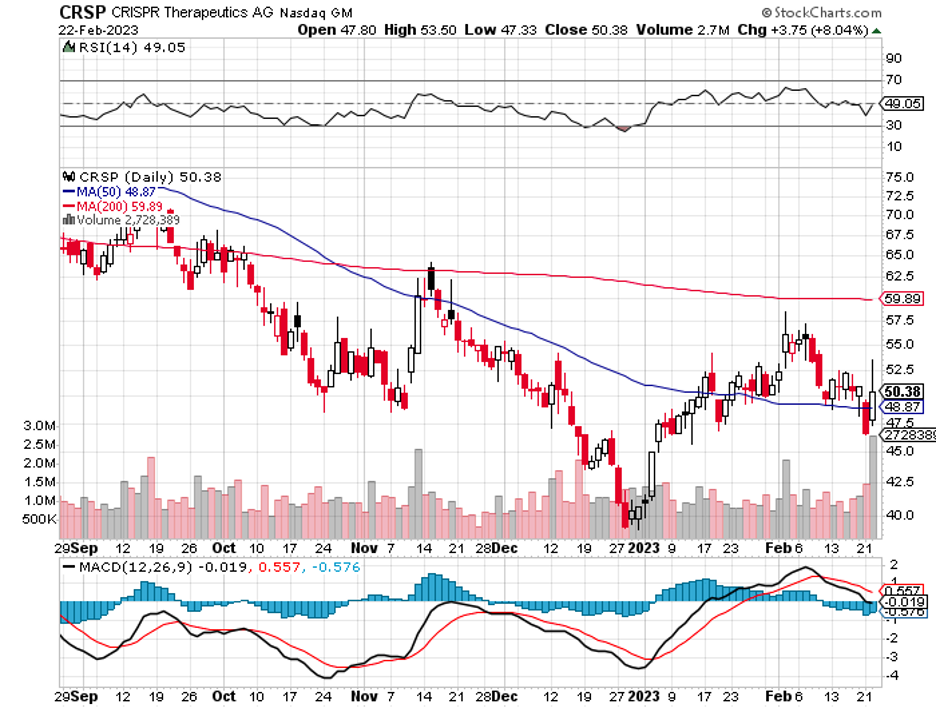
However, there is a field in the gene therapy world that has substantially rewarded investors: CRISPR gene editing.
CRISPR means Clustered, Regularly Interspaced Short Palindromic Repeats, which was discovered by Jenifer Doudna and Emannualle Charpentier. Their discovery won the Nobel Prize for Chemistry in 2020.
Basically, CRISPR is utilized by bacteria to recognize genetic sequences that belong to dangerous or harmful viruses and cleave them via specialized enzymes like CAS-9. Eventually, Doudna and Charpentier discovered that the system could be modified to target and remove, destroy, or even edit damaging genetic sequences in human beings.
This discovery gave birth to many biotech companies. Intellia Therapeutics (NTLA) was the brainchild of Doudna, while Charpentier co-founded CRISPR Therapeutics (CRSP).
Over the past five years, NTLA's share price has risen by 146% while CRISPR skyrocketed by 210%. In comparison, the S&P 500 recorded a 53% gain within the same timeframe.
Given the volatility of the field and market volatility, other CRISPR-centered companies failed to replicate this success.
The share price of Editas Medicine (EDIT) fell by 55% over the past five years. Caribou Biosciences (CRBU) also failed to ride the momentum and slid by 44%.
Still, there are positive updates amid the struggles of the sector.
The latest news is from CRISPR Therapeutics, which expects several catalysts in 2023 thanks to its promising pipeline of candidates and clinical trials. So far, one of the most anticipated catalysts is its biologics license application for its sickle cell disease candidate, which the company aims to file by March 2023.
CRISPR Therapeutics developed this candidate, called exa-cel, alongside Vertex Pharmaceuticals (VRTX). It would be the first-ever Crispr-based therapy to edit or rewrite faulty genes if approved. Based on the company’s data, patients who underwent this one-time treatment have continued to be free of sickle cell disease symptoms.
Every year, 100,000 patients in the US are reported to suffer from sickle cell disease. Many companies have offered treatments for this condition for years but no cure. Hence, CRISPR and Vertex’s one-and-done therapy has received a fast-tracked designation. Consequently, this would give the developers sought-after market exclusivity.
As anticipated, CRISPR Therapeutics’ competitors are hot on its heels with sickle cell disease treatments of their own. To date, Prime Medicine (PRME), Beam Therapeutics (BEAM), Editas, and Intellia have candidates queued for clinical trials.
Overall, the gene editing sector continues to be an exciting and interesting field. Investors looking to take part of the action in this segment should consider buying and holding CRISPR Therapeutics stock for at least five years because the company has a reasonable chance of becoming the most dominant name in the business soon.
Mad Hedge Biotech and Healthcare Letter
July 12, 2022
Fiat Lux
Featured Trade:
(THE LEADERSHIP BATON IS IN BIOTECH’S HANDS NOW)
(MRK), (SGEN), (CRSP), (VRTX), (BLUE), (BIIB), (LLY), (RHHBY)

Biotechnology companies have taken the reins and are expected to outperform the general market in the near future.
To date, the Nasdaq Biotech Index (NBI) has been up by 2.41% while the iShares Biotechnology (IBB) exchange-traded fund has climbed by 2.47%.
Numerous crucial factors place this industry in an advantageous position for growth. Alongside other segments of the pharmaceutical and healthcare industries, the biotechnology sector is essentially recession-proof.
With a roughly 40% fall in the biotech sector from 2021’s high, it’s highly likely for us to witness a boost in takeover activity in this space.
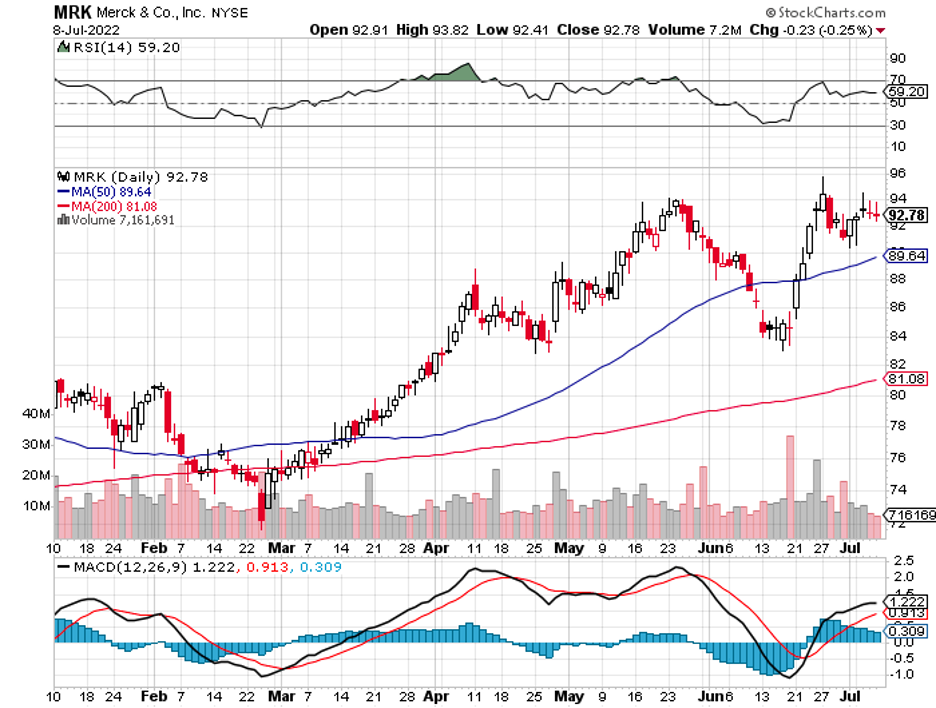
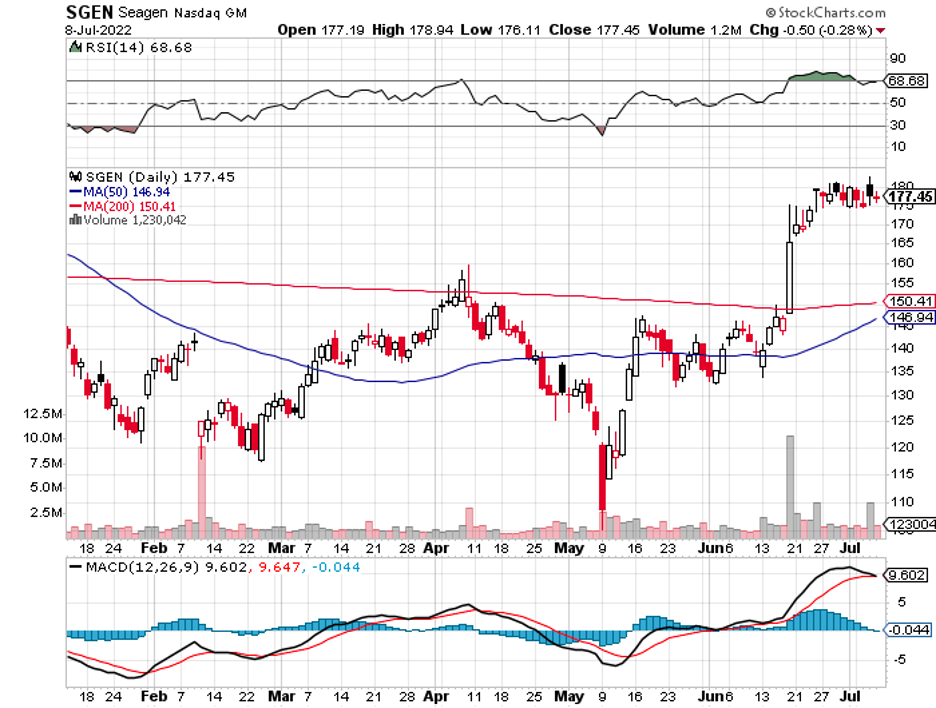
This is evident in recent reports of Merck (MRK) attempting to acquire cancer biotech Seagen (SGEN), as discussed in the June 30 issue of this biotech and healthcare letter.
The talks have progressed, and it appears that Merck is nearing the end of the process. The goal is to have the details worked out by the time the quarterly earnings report is released on July 28.
While no specifics have been made public, it is estimated that the larger healthcare company will pay a staggering $40 billion for this Seagen acquisition.
If this goes through, Merck will pay more than $200 per share for Seagen.
The news of this acquisition bolstered Seagen’s business as the stock rose by 4.6% at the time of the announcement.
This is welcome news given the perceived slowdown in biotech M&A activity since 2020. As a result, the idea fueled pessimism among investors who failed to see the big picture during this time period.
Analysis of 101 contracts signed by small, medium, and large biotechnology companies between January 2015 and June 2022 reveals that this year's contract volume and size are comparable to those of previous years.
In fact, there have been $17.7 billion in transactions so far in 2022. This translates to more than $13.9 billion in 2020 and $7.2 billion in 2021.
Some investors may be concerned about the quality of these acquisitions.
Even though the companies involved paid good premiums, the last 12 months' acquisitions were done at a big discount to the highest share prices of the businesses being bought.
To put it simply, there has been a problem with pricing in the sector as of late.
This is admittedly a "bittersweet" reality of recent biotech M&A transactions. As a result, market perceptions are clouded and investors are misled into believing that a much larger problem is brewing in the sector.
Executing megadeals is an obvious solution. This is why the Merck-Seagen merger is such good news for the industry.
The impact of this report suggests that large-scale M&A could be part of the path to the biotech sector's recovery.
The mere possibility of this transaction has already increased the SPDR S&P Biotech exchange-traded fund by approximately 20%.
In addition to Merck and Seagen, other biotechnology companies have been widely discussed as potential acquisition targets.
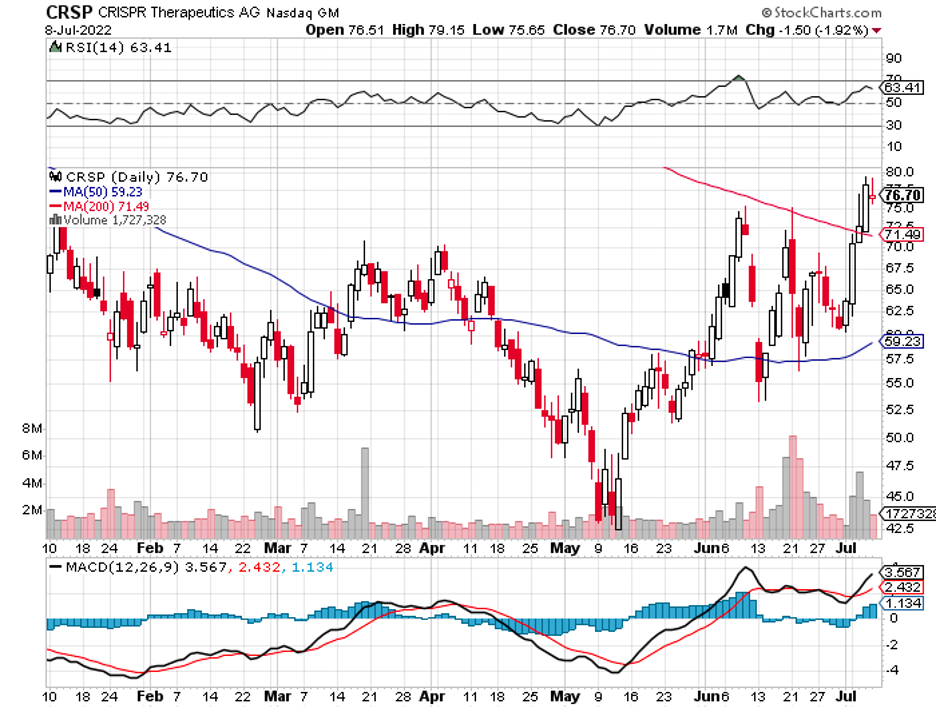
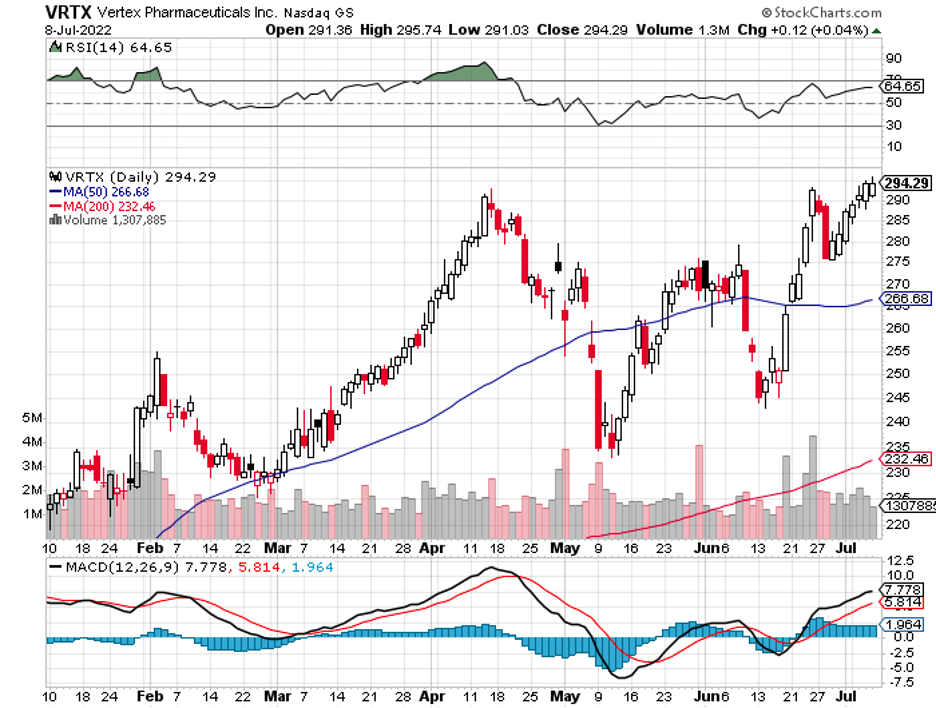
CRISPR Therapeutics (CRSP), which has a long-term partnership with Vertex Pharmaceuticals (VRTX), is a fan favorite. By the fourth quarter of 2022, the two intend to submit their sickle cell and beta-thalassemia treatment for approval.
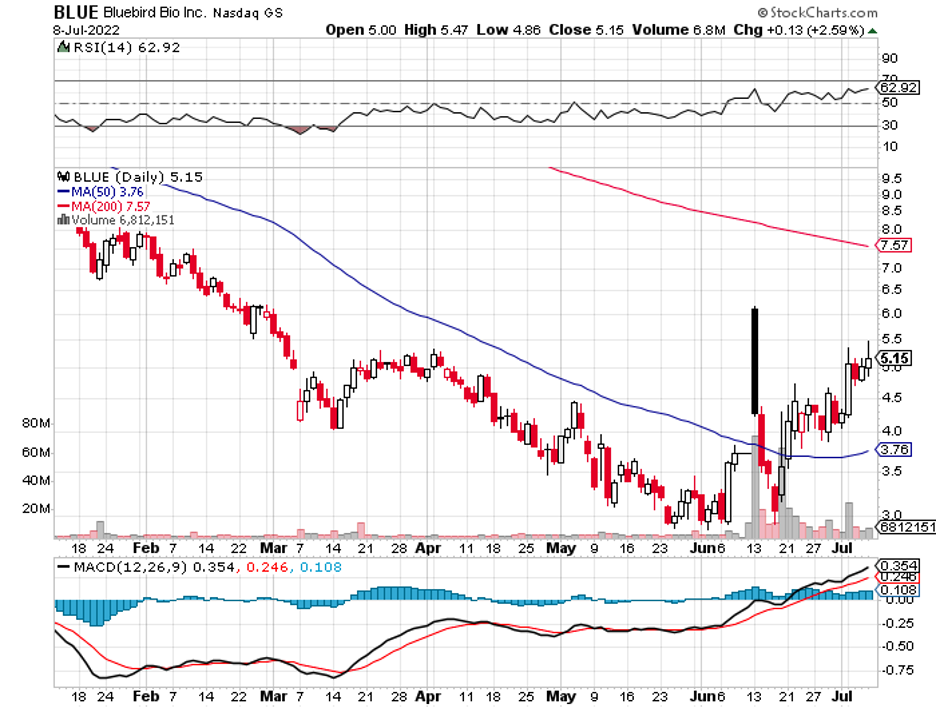
Bluebird Bio (BLUE) is another company that has been on the radar whenever acquisition discussions begin.
This gene therapy and cancer biotech has been unnerving investors for months, even before the pandemic triggered an economic crisis, due to its lackluster performance. Despite this, its gene-editing program has enormous potential.
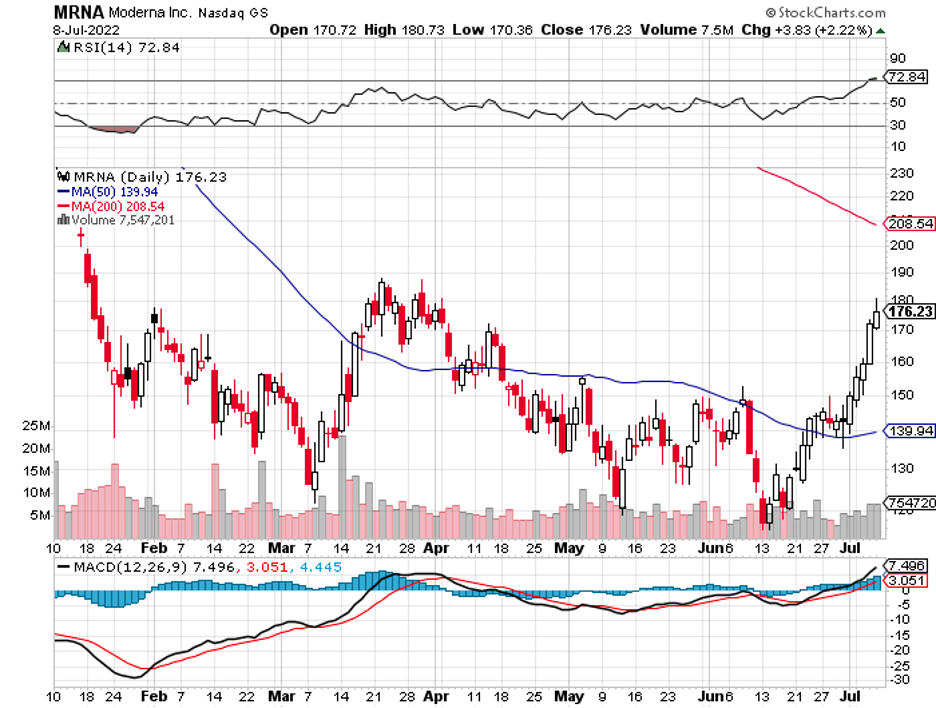
With a market capitalization of $368 million, it is an ideal candidate for Merck and even Moderna (MRNA). After all, both have been considering expanding its oncology program, and a dirt-cheap acquisition target appears to be an appealing option.
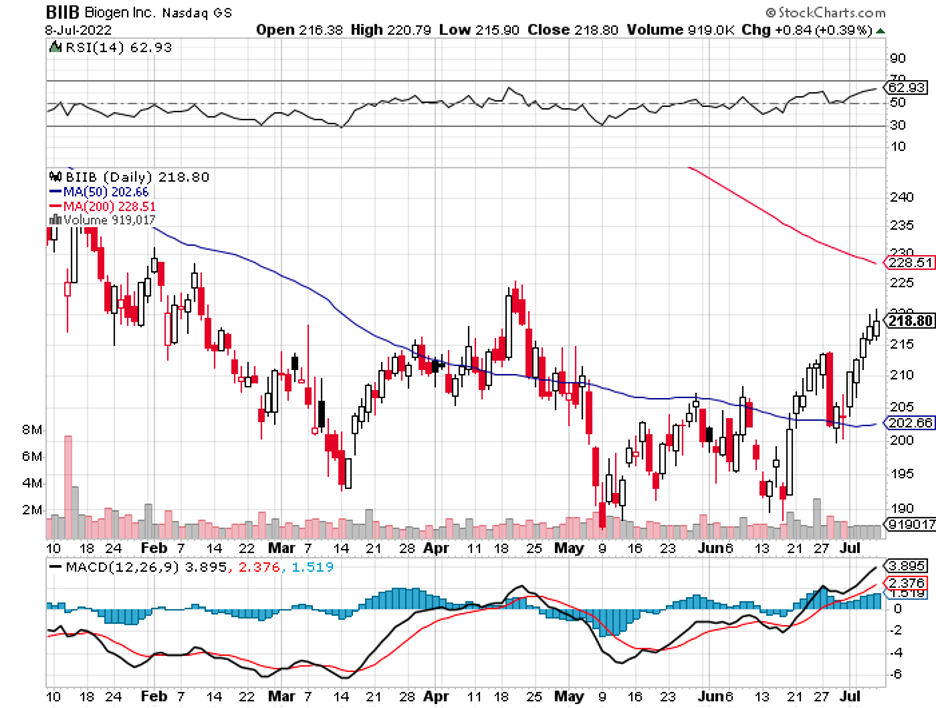
Biogen is another name associated with multiple interested parties (BIIB). Since its Alzheimer's treatment failed to materialize and deliver despite the biotechnology company exhausting virtually all available options to salvage the situation, the stock has yet to exhibit any signs of recovery.
After betting the farm on this candidate, Biogen has struggled to maintain its financial stability. In an effort to improve its cash flow and pay off its debts, the company has also been working overtime to advance the other programs in its pipeline.
Eli Lilly (LLY) and Roche (RHHBY), which have been working on their own Alzheimer's treatment, have recently been linked to Biogen.
With a market capitalization of $32 billion and a money-losing program, however, any transaction involving this biotech would require significantly more time.
Overall, it appears that biotechs are gradually regaining their footing. It is only a matter of time before all the pieces fall into place and the sector begins to move forward with full force.
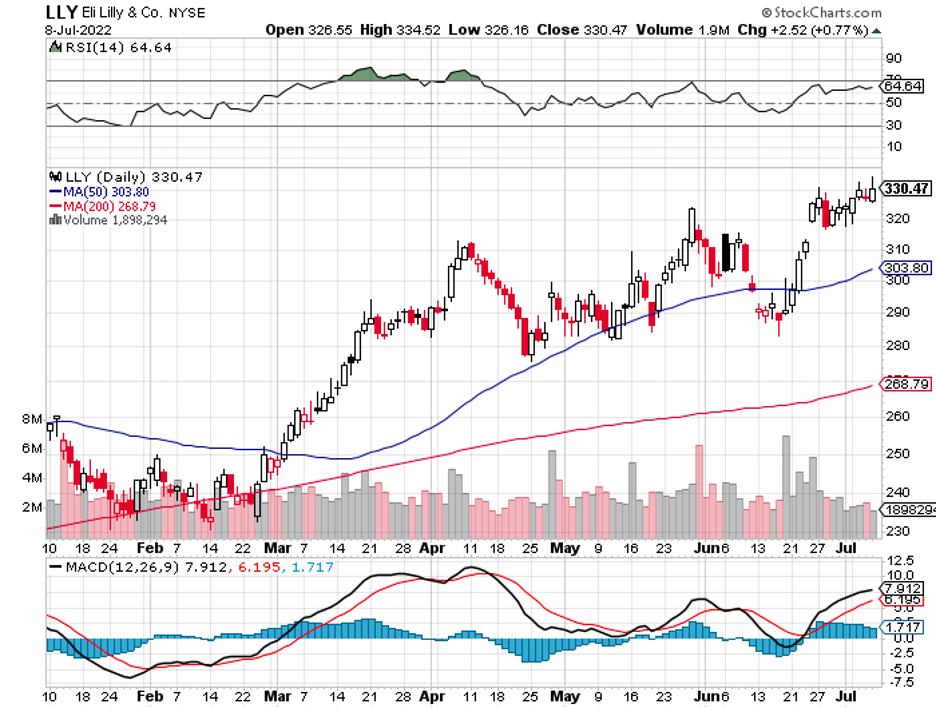
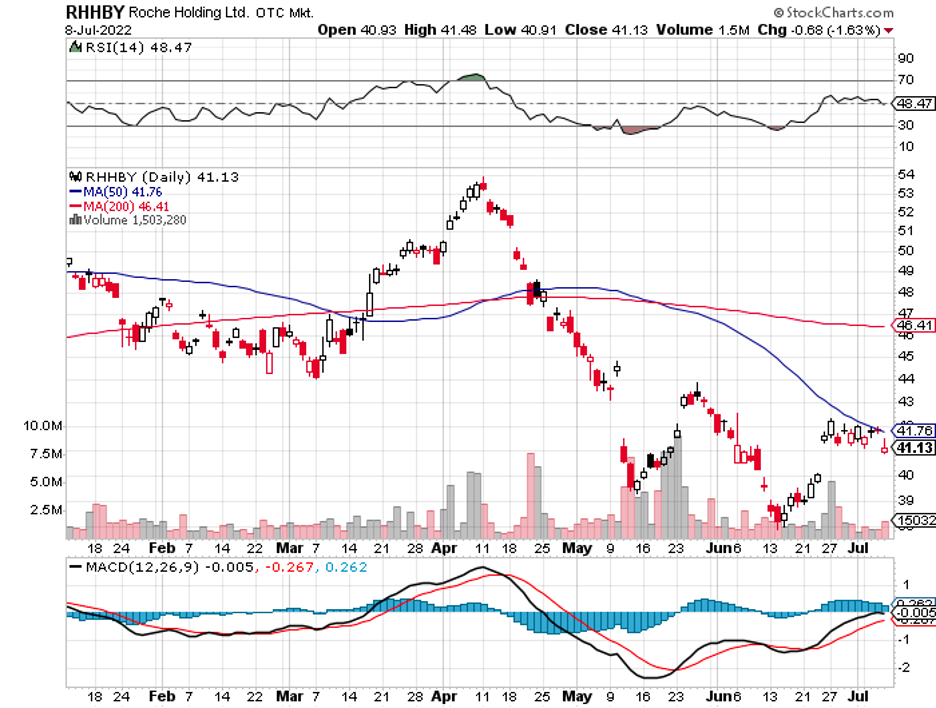
Mad Hedge Biotech and Healthcare Letter
April 14, 2022
Fiat Lux
Featured Trade:
(A BIOPHARMA STOCK BENT ON REDEMPTION)
(MRK), (BMY), (ABBV), (ORGN), (PFE), (VTRS), (MRNA), (BNTX), (CRSP), (VRTX), (BLUE), (BIIB)

It looks like we’re about to bear witness to a redemption journey.
Once upon a time, Merck (MRK) was a major player in the cardiovascular sector. Over the years, it has gradually diminished to a minor league name.
However, Merck has plans to reverse this fortune and reclaim its dominance in the cardio market. To date, it has eight new drug approvals and a slew of expanded labels queued in the next couple of years.
This decision is evident in Merck’s move to outbid Bristol Myers Squibb (BMY) in the auction for Acceleron Pharma, shelling out a whopping $11.5 billion to boost its cardio pipeline considerably.
While the deal may seem like a massive risk, Merck is confident that this deal holds the potential to open up the path to single-product peak sales reaching $10 billion by the mid-2030s.
In fact, there’s no need to wait for long to see some solid proof of Merck’s multibillion-dollar bet, as Acceleron already has a candidate set to be put on display by the end of 2022 or early 2023.
This Acceleron acquisition forms part of the “New Merck” touted when the company welcomed a new CEO and came on the heels of the success of the leadership that brought the mega-blockbuster cancer drug Keytruda.
It also signifies Merck’s conscious efforts to ease their heavily criticized over-dependence on Keytruda.
While the drug will lose patent protection after 2028, Keytruda still holds a significant portion of Merck’s sales. The treatment accounted for roughly 35% of the company’s total revenues last year.
The patent loss of a significant moneymaker is a typical problem for virtually every Big Pharma company, with AbbVie (ABBV) and Bristol Myers Squibb coming to mind as the most recent examples.
The go-to solution to this is pursuing mega-money mergers: AbbVie acquired Allergan for $63 billion while Bristol splurged on Celgene at $74 billion.
This quickly bolsters the existing pipelines and portfolios of the companies and assuages the fear of investors over impending revenue losses.
Instead of following this pattern, Merck did the opposite in 2021.
The company decided to downsize and established a spinoff segment: Organon (ORGN). The idea is to offload its biosimilars and other legacy products to focus on its core strengths.
This is reminiscent of Pfizer’s (PFE) move to spin out its Upjohn unit and merge it with Mylan to form Viatris (VTRS).
This move looks to have worked well for Merck and Organon as it allowed the parent company to focus on its blockbuster brands.
For instance, Bridion recorded a 28% year-over-year rise in 2021 to reach $1.53 billion in sales, while ProQuad reported a 14% increase to hit $2.14 billion.
Meanwhile, Gardasil rose to an impressive 44% to contribute $5.7 billion.
Even Merck’s Animal Health sector grew by 18% to record $5.6 billion.
There’s also Keytruda, which is projected to become the highest-selling drug at $24.3 billion by 2026.
These are only some of the blockbuster products in Merck’s portfolio expected to continue increasing revenues this 2022.
In addition, the company expects at least $5 billion from its COVID-19 antiviral drug Molnupiravir.
Looking at the trajectory and growth of the pipeline and existing programs, Merck estimates an additional 17% increase in its year-on-year revenue in 2022 to reach $56.1 billion to $57.6 billion.
Despite the move to establish a spinoff unit, the Acceleron deal hints at the possibility that Merck might be shifting to an open checkbook strategy.
Considering how relentlessly it pursued the deal, there’s a chance that the company would be at the bargaining table for a while in search of ways to protect itself against the pending Keytruda patent loss.
Some contenders for a potentially splashy offer from Merck are Moderna (MRNA) and BioNTech (BNTX), which could bolster the bigger company’s mRNA pipeline.
It can also splurge on gene therapy experts by targeting CRISPR Therapeutics (CRSP) and even Vertex (VRTX).
However, given bluebird bio’s (BLUE) flailing performance as of late, this small biotech could very well be a contender for a bargain deal.
Speaking of discounted stocks, Biogen (BIIB) is also reportedly under consideration simply because of its deeply discounted price following its disastrous Alzheimer’s disease program.
Whatever move it makes, one thing is sure: Merck, with its $208 billion market capitalization, is in a healthy and stable place financially.
More importantly, it has an excellent product portfolio and an exciting pipeline.
It has shown remarkable growth in the past years and impressive efforts to secure a great future, making it a solid stock to buy and hold for a long time.
Mad Hedge Biotech and Healthcare Letter
March 15, 2022
Fiat Lux
Featured Trade:
(AN UNDER-APPRECIATED STOCK WITH A BOATLOAD OF CASH)
(BMY), (CRSP), (VRTX), (BLUE), (GILD), (NVS)

Warren Buffett is nothing but a dyed-in-the-wool type of investor. A key strategy in his success is to target companies with notably solid fundamental businesses but with shares trading at a bargain or at least a discount in relation to their intrinsic value.
Needless to say, this value-oriented tactic has worked well for roughly six decades, with Berkshire’s stock delivering total returns of 6,450% on its capital.
Taking a cue from the Oracle of Omaha’s playbook, let’s take a look at one of the cheapest biotechnology and healthcare stocks in Berkshire’s portfolio to date and see how it has been performing.
At a hair below eight times its forward earnings, Bristol Myers Squibb (BMY) comes out as Berkshire’s third-cheapest stock holding overall.
This pharmaceutical giant, which has a market capitalization of $144 billion, is the sixth-biggest in the list of what is informally called the “Big 8” US pharma firms.
However, BMY’s stock had fallen by -2% in the past 12 months after experiencing some genuine momentum in 2021 when it reached $69 in August. The share price fell to $54 in December. It has since recovered and is now at $65.
A primary reason for investors snubbing this pharmaceutical giant is the impending loss of market exclusivity of three of its best-selling treatments, Revlimid, Opdivo, and Eliquis.
Although it’s reasonable to be anxious over these patent expirations, BMY has developed a great plan to not simply offset the future decline in sales but also to sustain the momentum of its top line all the way until 2030.
Basically, BMY has lined up multiple new drug launches spaced in the following years, with a number of these candidates expected to become potential blockbusters.
Another key part of the company’s growth strategy is acquisitions.
One of the significant moves BMY executed in recent years is its whopping $74 billion acquisition of Celgene in 2019, which is expected to bolster its immunology and oncology sectors. This was immediately followed by a $13 billion buyout of MyoKardia in 2020, which would expand its cardiovascular roster.
Celgene's deal granted BMY a valuable collection of pipeline assets, which the company has been leveraging in preparation for the patent cliffs.
Aside from the added $15 billion in annual revenue stream from Revlimid, which BMY used to boost its cash flow and pay off some debts, the company also inherited Reblozyl.
Since the acquisition, Reblozyl has gained approval for beta-thalassemia and anemia patients.
While this is not as groundbreaking as the gene therapies offered by CRISPR Therapeutics (CRSP), Vertex (VRTX), and even bluebird bio (BLUE), this treatment can still reach peak sales of $2 to $4 billion annually.
Another Celgene candidate poised to become an additional revenue stream for BMY is Inrebic, a JAK2 inhibitor created for myelofibrosis and polycythemia vera. This is projected to rake in $400 million in peak sales.
Zeposia, a treatment for autoimmune conditions, has already gained approval for multiple sclerosis and is queued for clinical trials for Crohn’s disease and ulcerative colitis.
If it receives the green light for all three, this is another $3 billion opportunity for BMY.
The inherited assets from Celgene are Breyanzi, a CAR-T therapy approved for large B-cell lymphoma, and Abecma, which is also an approved treatment for multiple myeloma.
These last two treatments are potential blockbusters as well.
Breyanzi’s list price is $410,000, with the therapy estimated to reach $3 billion in peak sales. Meanwhile, Abecma is listed at $491,500 and is projected to peak at $1 billion.
By 2029, BMY expects to develop new revenue streams worth $25 billion from its current portfolio and growing assets.
Looking at the above assets, BMY’s strategy becomes evident.
When BMY acquired Celgene for an exorbitant amount three years ago, the bigger company’s management team showed just how prepared they were to take the hit in the form of substantial debts in exchange for massive steps forward.
Adding to its expansion efforts, BMY has recently completed a deal with Century Therapeutics.
This marks BMY’s major foray into the promising cell therapy space.
While BMY has not concentrated on this sector before, it already has promising candidates in the form of its Celgene assets, Breyanzi and Abecma.
So far, Century and BMY have agreed to develop four different CAR-T cancer therapies on top of expanding the indications for Breyanzi and Abecma.
At the moment, the big pharma names focusing on this sector include Gilead Sciences (GILD) and Novartis (NVS).
This means BMY has a fighting chance to dominate in this market following its strategic collaboration with Century.
If all goes according to plan, BMY’s work with this cell therapy company might even turn out to be as lucrative as its deal with Celgene acquisition.
Overall, BMY has proven itself to be a reliable money-making titan in the biotechnology and healthcare industry.
BMY is a growth machine that consistently comes up with ingenious plans to grow over the years.
From $20.8 billion in 2017, its profits skyrocketed to an impressive $46.4 billion in 2021, indicating a remarkable 123% increase.
Moreover, the company anticipates that its free cash flow will surpass $50 billion by 2024, implying that it’s not worried over the impending loss of patent exclusivities and flexing its ability to generate a boatload of cash to complete even more collaborations and acquisitions.
Mad Hedge Biotech and Healthcare Letter
March 10, 2022
Fiat Lux
Featured Trade:
(COULD THIS BE THE NEXT BIOTECH BUYOUT CANDIDATE?)
(BLUE), (AZN), (ABBV), (BMY), (VRTX), (CRSP)

What is the common denominator of giant drugmakers AstraZeneca (AZN), AbbVie (ABBV), and Bristol Myers Squibb (BMY)?
Aside from being three of the biggest healthcare companies across the globe, all three have also completed high-profile acquisitions amid the pandemic.
AstraZeneca acquired Alexion Pharmaceuticals for $39 billion in December 2020.
Meanwhile, AbbVie wrapped up its whopping $63 billion acquisition of Allergan in May 2020.
As for BMY, this biopharma titan followed its jaw-dropping $74 billion acquisition of Celgene with a $13 billion merger with MyoKordia.
Since then, these deals have bolstered the lineups and deepened the pipelines of all three drugmakers, helping them secure their dominance in the healthcare space.
As for the acquirees, they also benefited from the transactions, particularly those struggling to get through tough situations prior to getting bought out.
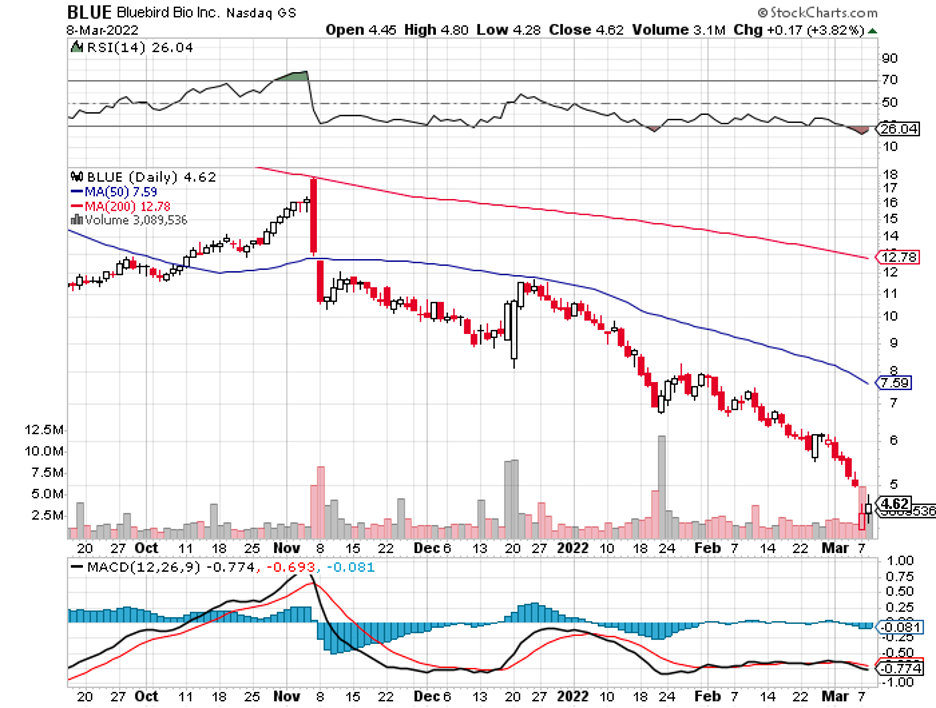
With that in mind, it looks like the biotechnology and healthcare sector has another potential high-profile acquisition candidate: Bluebird Bio (BLUE).
Bluebird Bio has recently fallen from investors’ grace following multiple setbacks.
The biotechnology company, once considered a trail-blazer in the gene therapy space, now finds itself without a CFO and left behind by competitors.
Its peers, who were initially eons away in terms of pipeline development, have figured out ways to work around Bluebird’s patents and even managed to outpace the company in launching new gene therapies to market.
Given all these factors, it is no surprise that investing in Bluebird bio has become synonymous with a recycler searching for value in random scraps and parts.
Over the last three years, Bluebird Bio’s shares have plummeted by more than 90%. From a $1.39 billion market capitalization, it is now at roughly $330 million.
That is a horrible performance based on any metric.
Bluebird bio has faced several headwinds that caused its stocks to fall apart.
One problem is the delay in the company’s Biologics License Application in the US for its transfusion-dependent beta-thalassemia treatment, LentiGlobin.
Bluebird Bio initially planned to complete this rare blood disorder therapy’s application by the second half of 2020. However, the company failed to submit some information requested by the FDA.
Another LentiGlobin-related issue is the temporary pause on the clinical trial for sickle cell disease treatment. Eventually, the suspension was lifted, but not before investors scurried away from the stock, following back-to-back concerns over the same treatment.
Other aggravating factors include Bluebird’s move to exit the European market following disagreements over the pricing of some of its gene therapies.
These issues saw Bluebird’s market cap sink, positioning it lower than rival gene-editing companies today.
Needless to say, this deeply discounted value could attract a bigger and expanding biopharma seeking to dip its toes in the gene-editing space.
While Bluebird might be struggling these days, it remains a promising company thanks to its candidates.
This becomes even more exciting since the company announced its plans to concentrate on severe genetic diseases. Although this is a small niche, there’s massive potential in this market.
A strong candidate in its roster is Zynteglo, which gained regulatory approval in 2019 and has yet to reach blockbuster status.
Patients with beta-thalassaemia normally have no other choice but to get blood transfusions regularly. Zynteglo drastically challenges this standard by offering a one-time curative treatment. In fact, saying that this is a life-changing breakthrough is an understatement.
Another potential blockbuster is Skysona, a treatment for a pediatric neurodegenerative disorder called cerebral adrenoleukodystrophy. This neurological disease is extremely rare, affecting only 50 patients in the US annually.
As for its pipeline, Bluebird has three major candidates nearing FDA approval in the US. This means 2022 and 2023 will be critical years for the company.
The first product is Lenti-D, which is similar to Skysona. If things go according to plan, then this treatment might receive the green light by August 2022.
Another product is Beti-cel, which was initially launched as Zynteglo. When this successfully penetrates the US market, this first-ever gene therapy option for beta-thalassemia will rake in roughly $1.87 billion by 2024.
Considering the potential of this market, Beti-cel inevitably finds itself facing off strong competitors in the space.
Thus far, the strongest is Vertex Pharmaceuticals (VRTX), which recently announced an additional $900 million investment in its collaboration with CRISPR Therapeutics (CRSP).
The third candidate is Lovo-cel, which is a sickle cell disease treatment.
This could be a major product for Bluebird, given the over 100,000 patients in the US alone that the company can target.
The goal is to finish the validation process by 2022 and submit Lovo-cel for approval by the first quarter of 2023.
Outside its pipeline candidates and approved products, Bluebird's manageable debt is another thing that makes it attractive for a potential buyout.
After all, cash is king, especially when it comes to biotechnology companies.
At the moment, Bluebird still holds roughly $442 million in the bank, and $46 million of this is restricted.
This indicates unrestricted liquid assets of approximately $396 million—an amount higher than its current market cap.
Consequently, this will allow Bluebird to comfortably weather at least the rest of the year until 2023.
It possesses a relatively solid secure position to hold itself together until its pending candidates start raking in revenues on their own.
Admittedly, Bluebird Bio has had several challenging years. There will still be uncertainties ahead, but it’s undeniable just how promising the company is at this point.
Overall, this stock is worth serious consideration, particularly for companies looking to get a head start in the gene-editing sector.
Legal Disclaimer
There is a very high degree of risk involved in trading. Past results are not indicative of future returns. MadHedgeFundTrader.com and all individuals affiliated with this site assume no responsibilities for your trading and investment results. The indicators, strategies, columns, articles and all other features are for educational purposes only and should not be construed as investment advice. Information for futures trading observations are obtained from sources believed to be reliable, but we do not warrant its completeness or accuracy, or warrant any results from the use of the information. Your use of the trading observations is entirely at your own risk and it is your sole responsibility to evaluate the accuracy, completeness and usefulness of the information. You must assess the risk of any trade with your broker and make your own independent decisions regarding any securities mentioned herein. Affiliates of MadHedgeFundTrader.com may have a position or effect transactions in the securities described herein (or options thereon) and/or otherwise employ trading strategies that may be consistent or inconsistent with the provided strategies.
This site uses cookies. By continuing to browse the site, you are agreeing to our use of cookies.
OKLearn moreWe may request cookies to be set on your device. We use cookies to let us know when you visit our websites, how you interact with us, to enrich your user experience, and to customize your relationship with our website.
Click on the different category headings to find out more. You can also change some of your preferences. Note that blocking some types of cookies may impact your experience on our websites and the services we are able to offer.
These cookies are strictly necessary to provide you with services available through our website and to use some of its features.
Because these cookies are strictly necessary to deliver the website, refuseing them will have impact how our site functions. You always can block or delete cookies by changing your browser settings and force blocking all cookies on this website. But this will always prompt you to accept/refuse cookies when revisiting our site.
We fully respect if you want to refuse cookies but to avoid asking you again and again kindly allow us to store a cookie for that. You are free to opt out any time or opt in for other cookies to get a better experience. If you refuse cookies we will remove all set cookies in our domain.
We provide you with a list of stored cookies on your computer in our domain so you can check what we stored. Due to security reasons we are not able to show or modify cookies from other domains. You can check these in your browser security settings.
These cookies collect information that is used either in aggregate form to help us understand how our website is being used or how effective our marketing campaigns are, or to help us customize our website and application for you in order to enhance your experience.
If you do not want that we track your visist to our site you can disable tracking in your browser here:
We also use different external services like Google Webfonts, Google Maps, and external Video providers. Since these providers may collect personal data like your IP address we allow you to block them here. Please be aware that this might heavily reduce the functionality and appearance of our site. Changes will take effect once you reload the page.
Google Webfont Settings:
Google Map Settings:
Vimeo and Youtube video embeds:
