Another year, another set of challenges and opportunities for investors. How can you make sure that you’re starting the year right?
If you’re looking to dip your toe in the biotechnology and healthcare sector, it won’t hurt to look at what the experts, such as Warren Buffett, are doing.
With a 55-year track record of 21% in annual returns for its Berkshire (BRK.B) investors, I’d say the Oracle of Omaha definitely knows his craft.
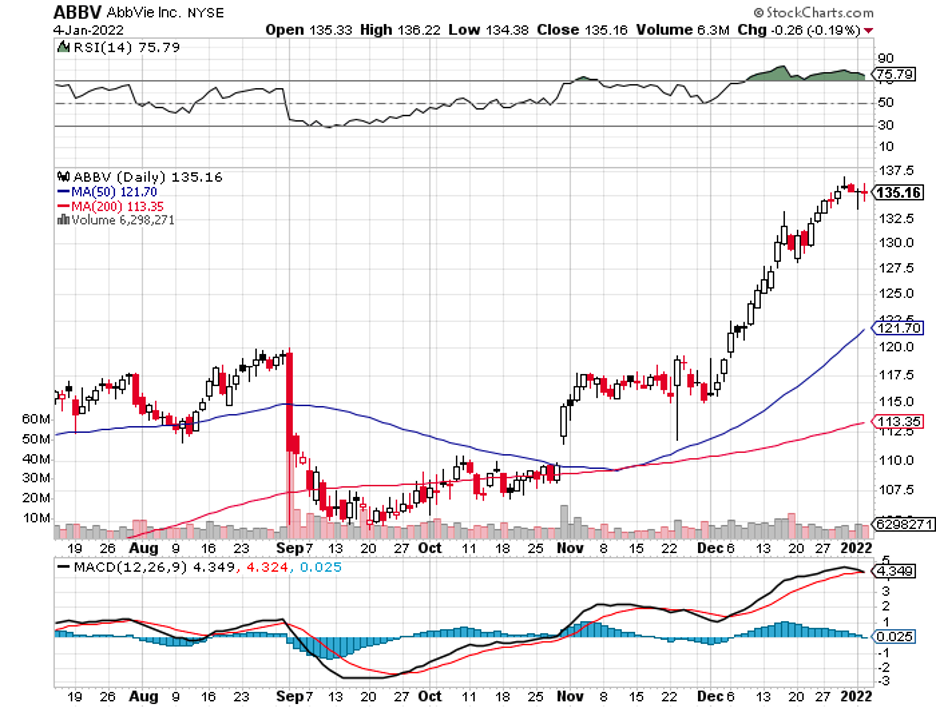
One of his most famous pieces of advice is to buy “wonderful companies at fair prices,” and I think this pearl of wisdom perfectly fits Buffett’s favored high-yield aristocrat: AbbVie (ABBV).
More interestingly, AbbVie is currently undervalued primarily due to overblown fears of patent loss for its top-selling drug Humira in 2023 and another in 2026 for Imbruvica.
Admittedly, the anxiety of investors is not entirely unfounded.
After all, AbbVie must replenish roughly $20 billion worth of annual revenue from Humira alone in the following years as the drug loses its patents and faces declining sales.
Obviously, losing revenue from a primary growth driver could be a massive problem for any company.
It could even lead to stagnating numbers in the next couple of years — a situation that the likes of Gilead Sciences (GILD), despite its $90.58 market capitalization, have become all too familiar.
Nevertheless, AbbVie isn’t simply twiddling its thumbs, waiting for the inevitable patent loss to happen.
The company has been busy preparing for the Humira and Imbruvica patent cliffs. In fact, it has been working to diversify its portfolio steadily.
One of the steps it undertook was to leverage its cash flow and $238.35 billion market capitalization to boost its R&D.
This led to AbbVie holding one of the industry’s most robust pipelines to date.
Some of the most promising products in its portfolio are Humira successors Skyrizi and Rinvoq.
Together, these two treatments are estimated to generate more than $15 billion in sales by 2025.
And AbbVie isn’t done yet.
Following its wildly successful formula in Humira, AbbVie is also looking into expanding the indications for the drug’s successors.
So far, Rinvoq has been able to deliver on this promise. Recently, this Humira successor has received FDA approval as a second-line treatment for psoriatic arthritis.
With this second indication, more and more investors are starting to believe that AbbVie has yet another blockbuster in the making.
Let’s look at the market for Rinvoq’s latest indication.
In the US alone, there are approximately 1 million to 2 million patients of psoriatic arthritis. For simplicity’s sake, let’s just say that there are 1.5 million patients in the US.
Generally, the first-line of treatment typically fails for 20% of psoriatic arthritis patients. That leads to roughly 300,000 patients who would be in need of second-line treatment.
For the sake of accuracy, it’s vital to point out that there are already a number of psoriatic arthritis treatments available in the US, such as Otezla and Enbrel from Amgen (AMGN). Moreover, JAK inhibitors are still facing potential restrictions from the FDA, thanks to the issue with Pfizer (PFE).
So, we can conservatively say that AbbVie’s Rinvoq might only be able to capture an estimated 7% of the psoriatic arthritis market share.
This translates to approximately 21,000. The annual list price for Rinvoq is at $63,000.
Taking into consideration the negotiation tactics of health insurers for price adjustments, the drug might go down to an annual list price of $44,000 instead.
Based on these conservative assumptions, Rinvoq would rake in sales of over $900 million—falling only slightly below the $1 billion blockbuster mark.
While this only hits less than 2% of AbbVie’s anticipated $56.2 billion annual revenue, this trajectory is a massive success for Rinvoq.
Bear in mind that this Humira successor has been on track to generate over $1.5 billion in sales in 2021.
Hence, adding $900 million from its psoriatic arthritis indication would offer a whopping more than 60% jump in its annual revenue.
Considering its history and trajectory, AbbVie is expected to continue to outshine its rivals in the industry.
Actually, the growth consensus for this stock is at 4% to 6.5%.
While that does not sound very impressive, it’s important to remember that the long-term growth rate for the whole industry is only 4%.
That easily puts AbbVie ahead of at least 63% of its peers in terms of growth.
Aside from the fact that this blue-chip stock is an excellent way to enjoy a solid 4.3% yield these days, the company is proving to be effective in ensuring that it delivers market-beating returns in the long run.
Needless to say, AbbVie qualifies as a classic “wonderful company at a fair price.”