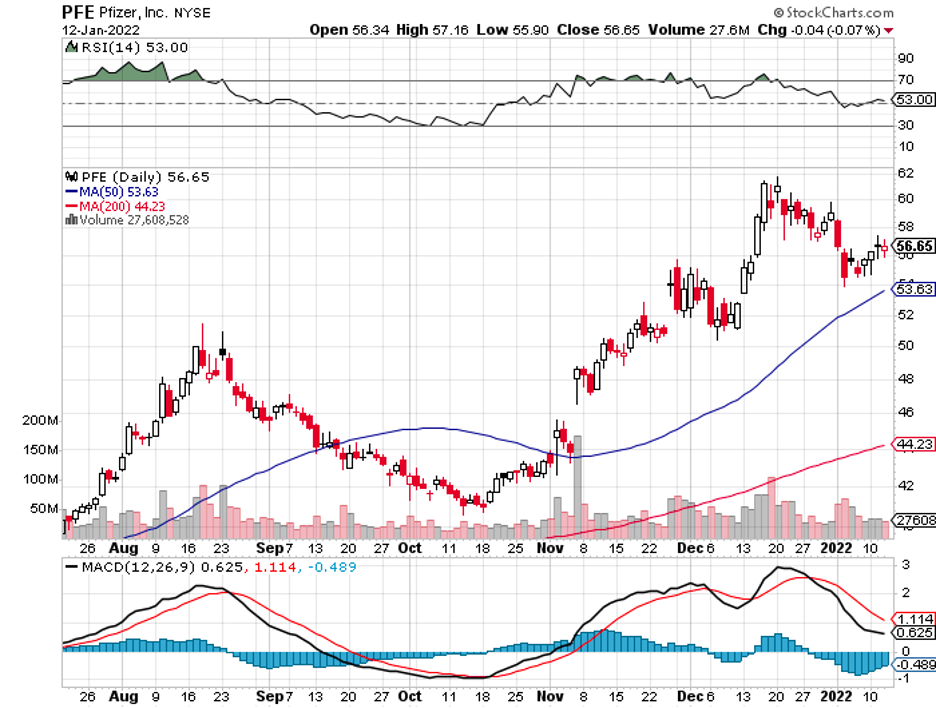
Amid the pandemic fatigue hounding everyone these days, one name continues to attack the situation with consistent vigor: Pfizer (PFE).
It’s not a stretch to say that its COVID franchise is the most popular line in Pfizer’s portfolio today.
Needless to say, this is highly lucrative from a shareholder’s point of view. The company’s vaccination business has recorded over 3 billion doses to generate roughly $36 billion in sales from Comirnaty alone in 2021.
Riding the momentum of its successful 2021, the company anticipates an even more successful 2022.
So far, Pfizer is targeting an increase in its Comirnaty production to hit at least 4 billion doses this year.
Aside from being one of the first companies to develop a vaccine, the company has also created a highly effective antiviral COVID treatment that can be taken orally: Paxlovid.
While Merck (MRK) has earlier announced its move to come up with a similar oral treatment, Pfizer’s pill proved to be more effective.
Actually, customers are starting to take note of the difference and are switching brands. France already canceled their agreement with Merck and decided to order Pfizer’s Paxlovid instead.
This once again underscored the dominance of Pfizer’s brilliant R&D segment and the company’s capacity to rapidly come up with highly effective solutions for issues involving COVID.
The way Pfizer has been handling the COVID situation can be compared to Roche’s (RHHBY) approach and eventual blockbuster success with Tamiflu over 20 years ago.
Although the flu is obviously not as deadly as the coronavirus, it still caused widespread economic breakdown and health problems.
When Tamiflu eventually entered the market, the world was finally granted a simple medical answer for what was initially thought to be an unsolvable health problem.
Pfizer’s Paxlovid could very well be the Tamiflu for COVID.
Looking at Paxlovid’s effect in terms of revenue, it’s safe to say that this oral treatment can drive medium-term growth for Pfizer.
To date, Pfizer disclosed that Paxlovid would be sold for roughly $700 for each treatment course.
Let’s use the US numbers as an example to help put things in perspective. So far, the country has recorded approximately 170,000 cases per day.
If we assume that this will be the average for 2022, then there will be about 62 million COVID patients this year.
Let’s say that only 40% of these patients qualify for Pfizer’s treatment; then this would reach 24 million people at $700 each to rake in roughly $17 billion in total revenue in the US alone.
The number would definitely be significantly higher considering that Paxlovid will be offered as a global COVID treatment.
It’s evident that Pfizer’s efforts are paying off, as the sheer earnings power of the company’s COVID-19 pandemic franchise could provide a medium-term boon for its investors.
In 2021, Pfizer recorded a 130% growth in its revenue, with the numbers still climbing.
While its pandemic response has become its primary growth driver, Pfizer’s other key segments also posted promising revenues.
To sustain its climb, the company has continued to invest in R&D heavily.
A notable investment it made recently is an $8 million upfront payment to Codex DNA (DNAY) for the smaller biotechnology company to “produce certain materials of interest to Pfizer.”
According to the deal involving the exclusive product, Codex expects $10 million in technical milestone payments, up to $60 million in clinical development milestones, and $180 million in sales milestones.
Codex DNA is a small biotechnology company with a market capitalization of $267 million. It’s a spinoff from a California company called Synthetic Genomics.
While Pfizer and Codex have yet to share their plans publicly, we can hypothesize that it has something to do with the large biopharma using the small biotech’s technology to accelerate its mRNA vaccine development process.
After all, Codex’s distinct value proposition lies in its rare ability to automate various elements of the entire process. Its push-button, end-to-end solutions promise to build functional grade synthetic mRNA and DNA.
In effect, this will save cost and time for its clients.
Aside from Pfizer, this small biotech has been collaborating with other organizations like Duke University and MIT.
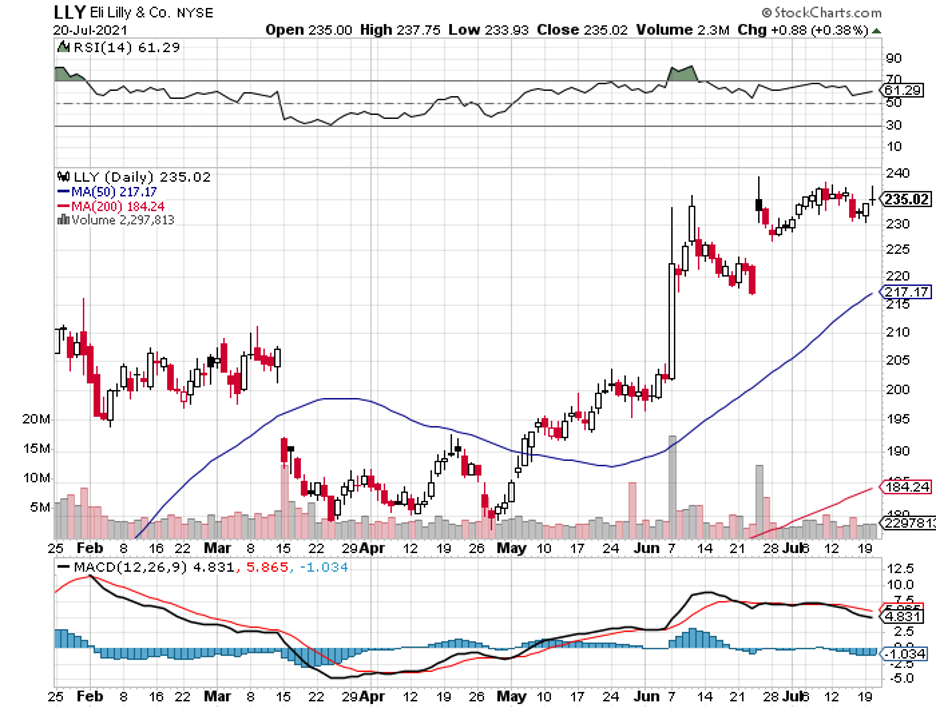
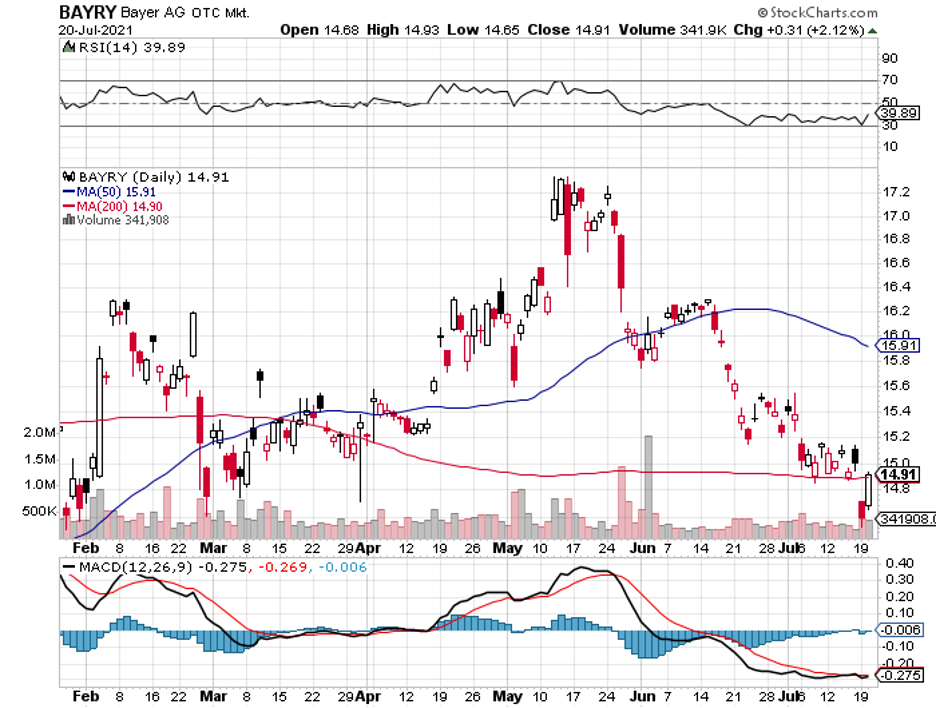
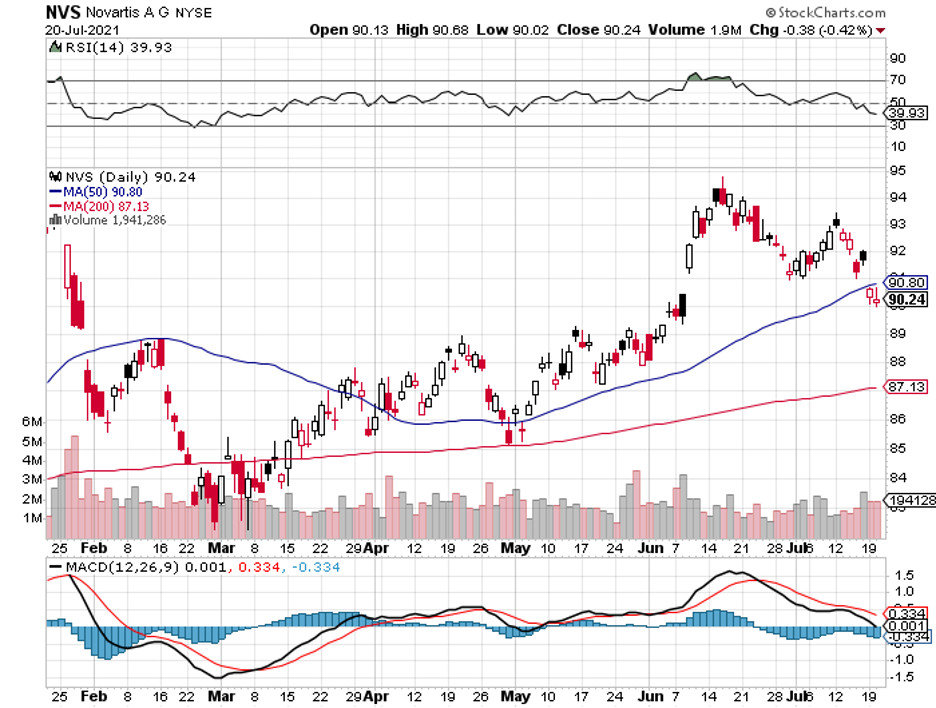
It has also been working with large biopharmas, including Johnson & Johnson (JNJ), Eli Lilly (LLY), BioNTech (BNTX), Merck, and even gene therapy expert Editas (EDIT).
For 2022, Pfizer is anticipated to generate at least $96 billion in sales, showing off a jaw-dropping 17.2% jump from its 2021 revenue and a 229% increase from 2020.
As we slowly accept that COVID will become a staple in our lives in the coming years, I think investors would be wise to add proven “experts” in their portfolio to take advantage of the ever-present and increasing demand.