This year has been challenging for the majority of the stocks, with even the strongest and most dominant names struggling to keep up. The three major indexes all slipped into bear territory while economic issues such as rising inflation brought turbulent earnings seasons across virtually every industry.
Still, 2022 has revealed a handful of exceptions. Some businesses delivered good news and, against all odds, solid stock performance. Some investors lined up to buy shares of these companies. While some have soared to unreasonable prices, it’s not too late to invest in other players sold at modest prices.
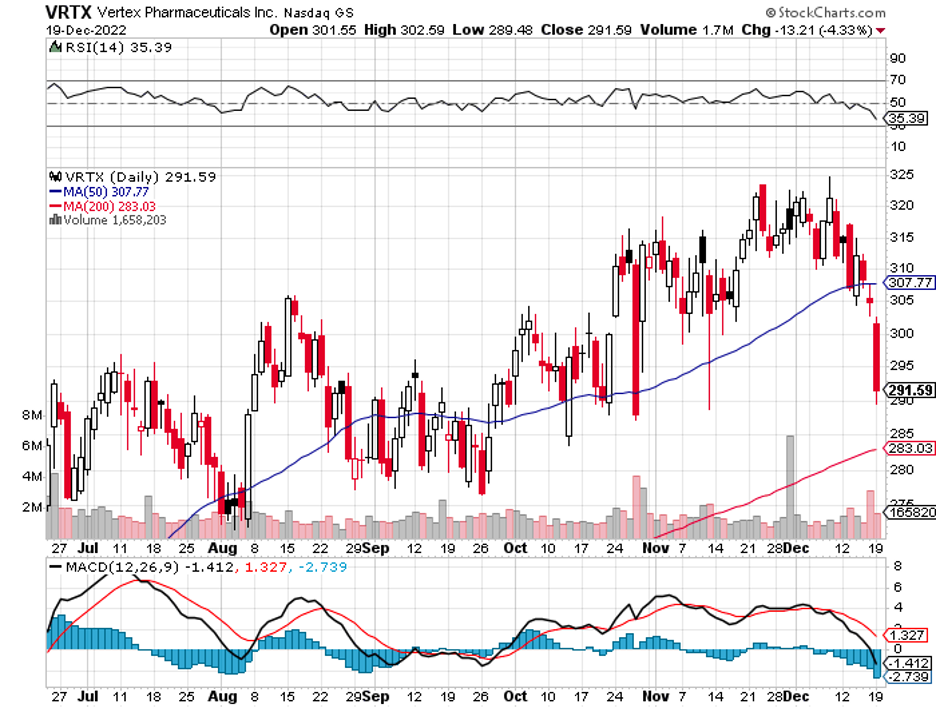
A particularly promising stock that meets these criteria is Vertex (VRTX).
Vertex has risen notably this year, recording a 38% boost to date. However, it’s trading at roughly 20 times its forward earnings predictions. Hence, buying this stock could very well guarantee solid investment in the long run. After all, the following years are expected to be filled with significant turning points.
An excellent starting point in reviewing Vertex’s potential is its portfolio. Right now, the company has six drugs sold commercially, reporting $7.5 billion in revenue last year. All six focus on cystic fibrosis (CF).
Vertex has been hailed as the worldwide leader in the CF market for years. On an even more promising note, the company is projected to sustain this momentum until the late 2030s.
Specifically, Vertex’s most recent CF treatment, Trikafta, has presented plenty of room for revenue growth in the years to come, courtesy of anticipated additional approvals in more countries and younger age brackets.
Vertex is also reviewing another CF candidate, which is now in Phase 3 trials. Based on previously released data, this new product has the potential to become even better than Trikafta.
Another CF candidate queued for review is the drug Vertex has been working on in collaboration with Moderna (MRNA). If approved, this product will cover patients not eligible for the current CF roster of Vertex.
Surprisingly, however, the potential catalyst for Vertex’s share price in the coming years has absolutely nothing to do with its highly successful and established CF program.
Rather, it has something to do with the company’s new venture on blood disorders: Exa-cel. This is a one-time cure developed by Vertex and Crispr Therapeutics (CRSP), which targets two blood orders. To this day, there remain minimal options for patients with these diseases.
For two key reasons, gaining approval for Exa-cel could be a massive game changer for Vertex. One is that it can provide definitive proof that the company can expand beyond its CF programs.
The second is that it would provide an additional revenue stream for Vertex, and that’s always a desired outcome regardless of the billions of dollars the company is already generating.
CF sales have clearly powered Vertex’s net income, which increased by about 1,140% in the past five years. With exa-cel, though, it’s evident that the company has been working to diversify its market to cover other diseases.
Reviewing its pipeline, Vertex has 18 programs with excellent chances of getting commercialized in the next 10 years. These run the gamut of treatments and therapies, including promising results for sickle cell disease, type 1 diabetes, kidney disease, and acute pain relief.
While it’s impossible to accurately determine the amount of money these drugs could make by 2032, it’s not that hyperbolic to believe that they can at least contribute several billions to the company.
Overall, Vertex stock offers a bright and solid future. In the next 10 years, the business would evolve into a much bigger, more entrenched, and more diversified entity. That means it would be a less risky investment compared to today.
Vertex would be an excellent choice for patient investors seeking to start a position in some biotechnology and healthcare companies.