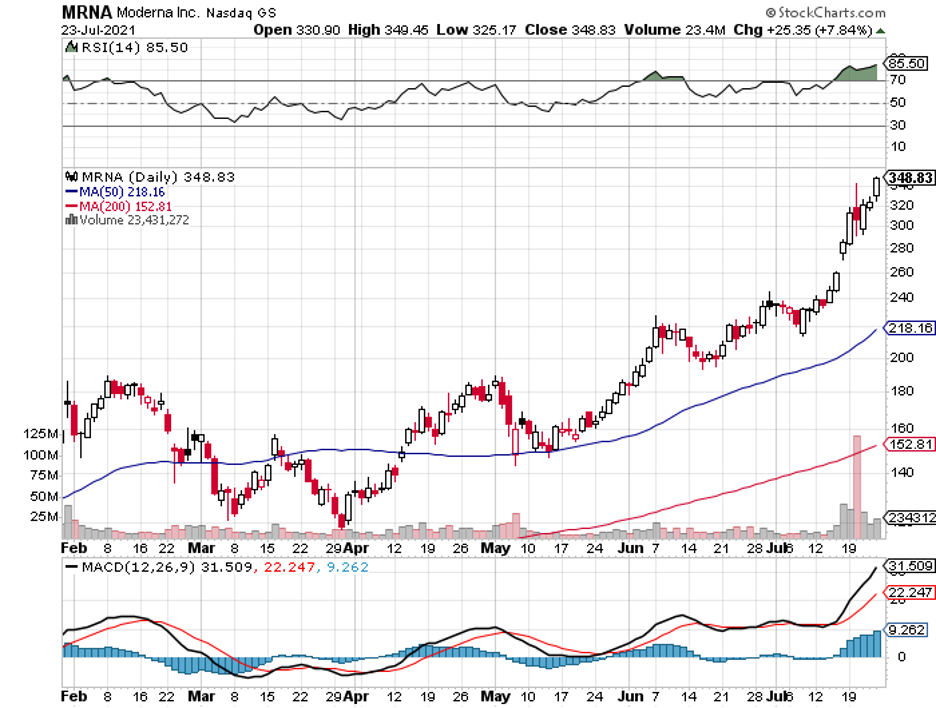
Many investors have amassed a fortune since the pandemic started in the early months of 2020 by betting on COVID-19 vaccine candidates. Moderna (MRNA), for example, has skyrocketed to over 1,500% since last year.
However, there was an even bigger winner: Novavax (NVAX).
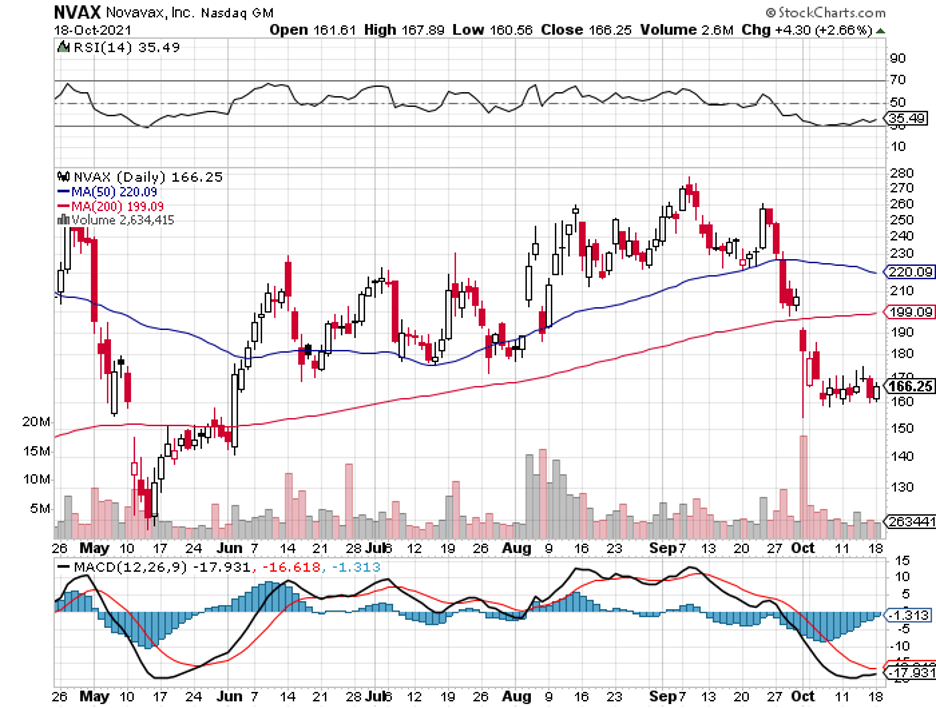
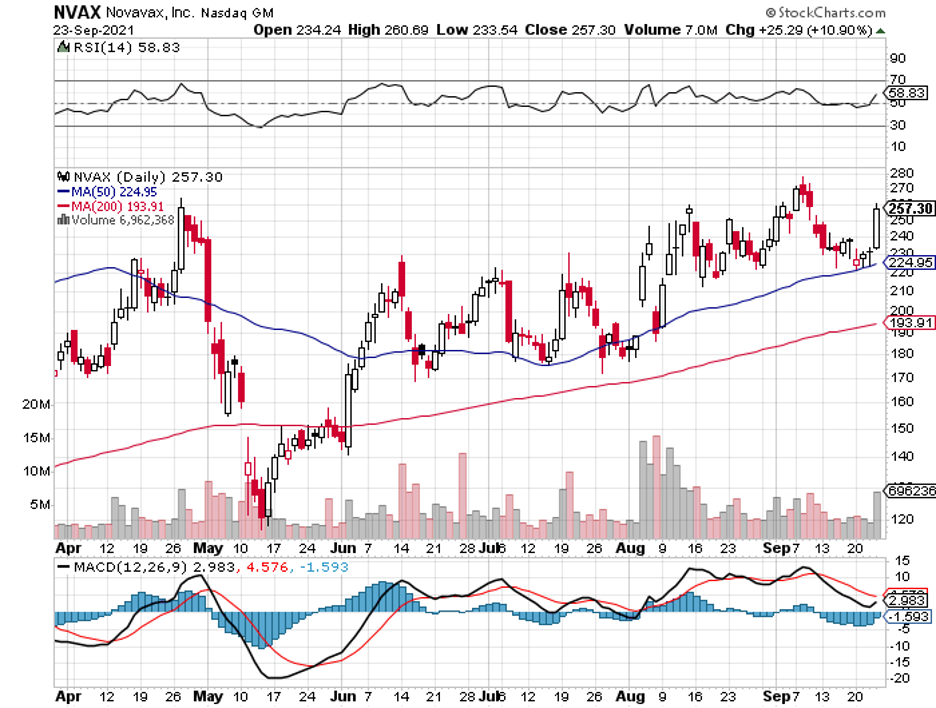
Novavax shares actually rose by a jaw-dropping 4,000% since the COVID-19 pandemic began, with the company’s wild rollercoaster ride still not reaching its end anytime soon.
In fact, Novavax has been quite volatile in 2021, rising by more than 180% in early February only to have most of those gains practically wiped out a mere three months after.
Then, the stock managed to show off a strong rebound over the following months. Since early September, though, Novavax’s share price has fallen by over 35%.
While these can be discouraging for some investors, I think that the befuddling gyrations that had us reeling in the past months tell a different story: We might have just discovered the biggest bargain among the leading COVID-19 vaccine stocks in the market today.
There are two possible reasons for the Novavax selloff recently.
The first is the company’s delayed filing of its own COVID-19 vaccine candidate, NVX-CoV2373. This is particularly frustrating considering that Novavax has failed to meet its deadlines multiple times now.
Nonetheless, I prefer to look at these delays as mere speed bumps than actual roadblocks that hinder the company from achieving its goal.
After all, the issue is not on the vaccine’s safety and efficacy—the results have been proven to be highly compelling—but on manufacturing concerns, which can eventually be resolved.
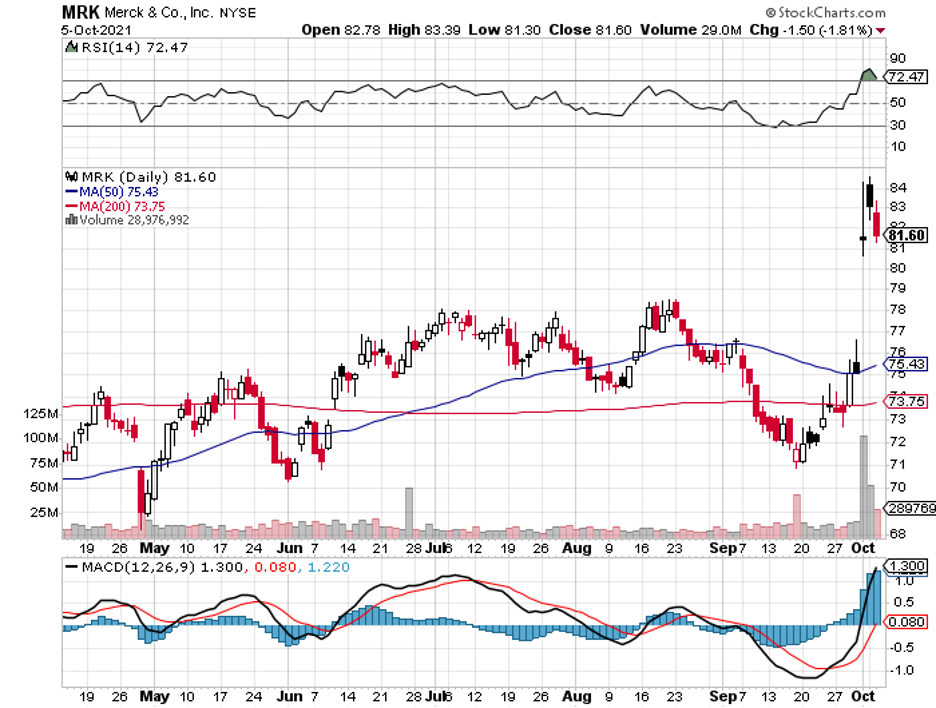
The second reason is the recent update from Merck (MRK) and its partner, Ridgeback Biotherapeutics, on their COVID-19 pill. Of the two, I find this reason to be an overreaction by the market.
None of the vaccine developers should ever be negatively affected by Merck’s oral treatment. While some people might choose not to get the vaccine if and when the pills become available, practically all governments worldwide are still committed to vaccinating their citizens.
Moreover, the COVID-19 vaccines will probably be necessary every year.
When Novavax gets the required authorizations, the company is set to generate a boatload of cash. The biotechnology company is anticipating to supply up to 200 million doses in the European Union alone.
The entire COVID-10 vaccine market is projected to be worth $115 billion by the end of 2021. Moderna is estimated to deliver up to 3 billion doses, while Novavax is expected to produce up to 2 billion doses by 2022.
The rest of the anticipated 14 billion doses will be divided among the other vaccine makers.
Among them, though, Novavax is expected to be the stand-out.
For one, its vaccine candidate appears to be the most robust and affordable at $16 per dose compared to Moderna’s $25.50 and Pfizer-BioNTech’s $22.80.
The growing number of reports on the side effects of mRNA vaccines, which are said to be of a higher rate than Johnson & Johnson’s (JNJ) candidate, can also be a turnoff for many people.
Since Novavax uses a more traditional and familiar vaccine technology—the same one used for the flu, HPV, and Hepatitis B—it causes lower side effects rates.
More importantly, these are mild symptoms like muscle pain and fatigue compared to the heart inflammation concerns raised among those jabbed with Moderna or Pfizer vaccines.
While the side effects from the other two occur in relatively small populations, Novavax is anticipated to be perceived as the more reassuring option, especially for people who are still uneasy with the new technology of mRNA vaccines.
More importantly, Novavax can offer a solution to the global problem of vaccine hesitancy for COVID-19.
To offer a context on how important this is, 48% of Russians and 27% of Americans refuse to take the vaccines.
As of September, only 181.2 million individuals in the US, or 55.1% of the country’s population, have agreed to be fully vaccinated. Needless to say, overcoming this hesitancy would be a massive relief for everyone.
Apart from its COVID-19 vaccine, Novavax has also been working on an influenza vaccine candidate, NanoFlu.
Recently, results from the NanoFlu clinical trials showed that it’s way more effective than the leading brand today, Sanofi’s (SNY) Fluzone Quadrivalent.
If NanoFlu gains approval, this will be another huge growth driver for Novavax.
To put it in perspective, Sanofi’s Fluzone generated $2.9 billion in sales in 2020—a market that Novavax can also tap into and might even dominate.
Meanwhile, the flu vaccine market in the US alone continues to expand.
From 2020 to 2021, the number of flu vaccines administered rose by 11% year over year to reach 193.8 million doses. That’s roughly 59% of the US population—a country that’s only 7th place in terms of the global rates of flu vaccination.
That signifies a colossal market opportunity worldwide for Novavax to capitalize.
At this point, Novavax has yet to secure any approval or official authorization for NVX-CoV2373 or NanoFlu—and that’s the best reason to add this stock to your portfolio.
Investors are given a chance to seize shares of the stock while it’s still in the stage where its most lucrative catalysts are just lurking around the corner.