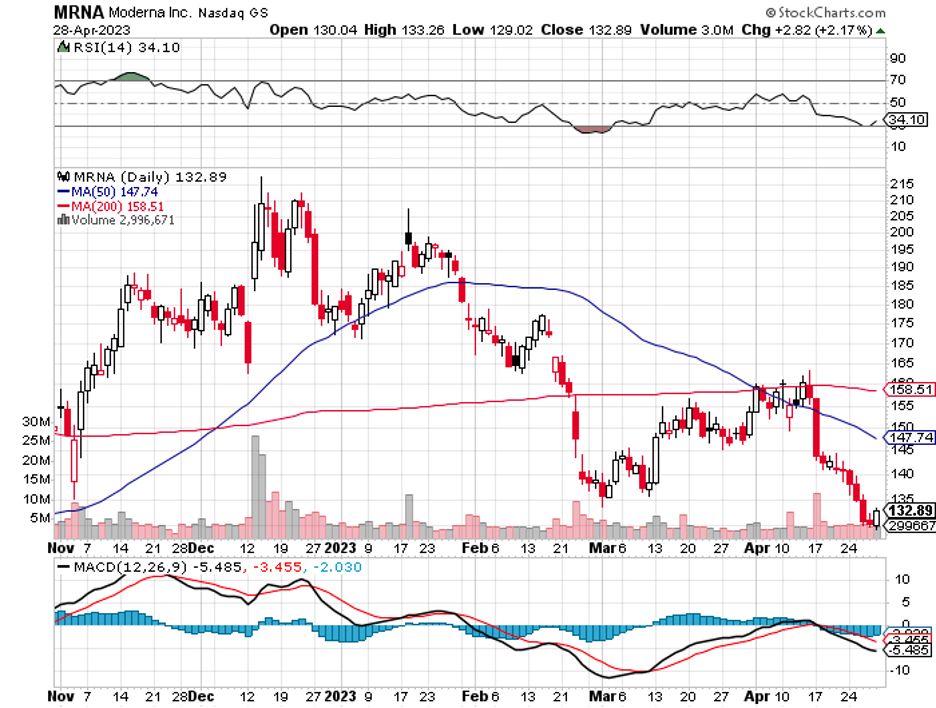
When you think of the pioneering biotech, Moderna (MRNA), artificial intelligence (AI) and quantum computing might not be the first things that come to mind. Instead, you might associate Moderna more with its work in traditional laboratory research and as a leading coronavirus vaccine manufacturer.
However, Moderna has taken significant strides into the realm of AI. In fact, the biotech utilized AI during the early stages of developing its coronavirus vaccine and has also implemented the technology for other business purposes.
Now, Moderna is taking things a step further by partnering with International Business Machines (IBM) to explore the potential of AI and quantum computing in enhancing its messenger RNA research.
Needless to say, this innovative collaboration could potentially revolutionize the biotech industry.
To understand Moderna's recent developments in AI and quantum computing, it's important to first have a grasp of its mRNA technology.
Unlike traditional vaccine production that involves growing viruses in a lab, Moderna produces mRNA that provides the body with instructions to treat or prevent a particular illness. This innovative process is already faster than traditional vaccine production methods. But AI has played a significant role in making the process even faster.
Moderna has been able to leverage AI and automation to scale up mRNA production significantly. In fact, the company's mRNA production for experiments went from about 30 per month to 1,000 per month thanks to AI. Additionally, AI has contributed to the generation of more effective mRNA sequence designs, saving researchers considerable time.
Let's now take a closer look at the implications of Moderna's partnership with IBM.
One of the primary areas of focus is IBM's generative AI for therapeutics, which has the potential to provide Moderna researchers with a deeper understanding of molecular behavior, facilitating the development of new molecules for therapeutics.
Moreover, IBM's expertise in quantum computing could prove invaluable in speeding up the discovery of new treatments, enabling Moderna to push the boundaries of medical research and improve patient outcomes.
Quantum computing differs from traditional computing in its use of a system that allows for states beyond the binary 1s and 0s. Quantum computers can understand information as 1, 0 or something in-between, offering the potential for individual bits to be in multiple states at the same time. This characteristic may be beneficial in modeling the dynamic interactions among drugs, enzymes, cells, and proteins that are continuously changing.
The use of advanced systems in molecular modeling has been challenging for earlier generations of hardware. However, the incorporation of quantum computing could revolutionize the way biotech companies solve these complex problems.
As a starting point, Moderna will be part of IBM's enterprise accelerator program, which provides a platform for "quantum curious" companies to invest in building their expertise in emerging areas. This program gives access to IBM's network of computing systems and specialized training on the use of quantum computing for life sciences research.
As part of this collaboration, Moderna will gain access to MoLFormer, a powerful AI model that can accurately predict a molecule's properties. This tool will prove particularly valuable in Moderna's efforts to improve the lipid nanoparticles that encapsulate its mRNA treatments.
Additionally, the partnership includes investments in generative AI programs that will assist in the design of innovative mRNA-based treatments and vaccines, helping Moderna to further cement its position as a leader in the biotech industry.
IBM had previously attempted to make a name for itself in AI-powered drug discovery, offering services through its Watson platform.
However, these offerings were ultimately discontinued in 2019. Despite once partnering with major names in cancer research such as Pfizer (PFE), Novartis (NVS), Illumina (ILMN), as well as Teva (TEVA) for drug repurposing, IBM has shifted its focus to other areas of the life sciences industry.
As quantum computing technology continues to evolve, however, its potential applications have begun to attract some of the biggest names in biotech.
Companies like Novo Nordisk (NVO), Roche (RHHBY), and Boehringer Ingelheim have partnered with industry giants like Google (GOOGL) to explore the possibilities of this cutting-edge field, which is quickly moving from the realm of science fiction into a scientific reality.
As for the question of whether these moves can be a game-changer for Moderna, the answer is likely yes.
Moderna has already experienced significant benefits from AI in its processes, both in and out of the lab. With access to IBM's platforms, there is potential for further improvements in the company's research and development of new treatments and vaccines.
Efficiency, speed, and precision are crucial factors in drug and vaccine development, and any improvement in these areas could have a significant impact on Moderna's success. Although the results of the IBM partnership may not be immediately visible, Moderna's investments in AI and quantum computing could pay off in the long run.
With continuous innovation and portfolio expansion, Moderna is well-positioned to capitalize on market opportunities presented by mRNA technology and achieve substantial revenue growth in the years ahead.
Therefore, investors should not be overly concerned about short-term stock price fluctuations or declines in revenue from coronavirus vaccines. After all, Moderna has a robust pipeline and has demonstrated significant potential with promising clinical trial results.
Hence, investors should consider Moderna as a long-term investment opportunity, making it a valuable addition to any investment portfolio.