The New Shot Caller
There’s nothing like a little competition to shake things up in the vaccine world, and it looks like Vaxcyte (PCVX) just lobbed a serious challenge at the reigning king of pneumococcal shots, Pfizer (PFE).
You know that satisfying smack when a basketball, held under water, bursts free and cracks you in the nose? That’s what’s happening in the vaccine market right now.
Vaxcyte just threw down the gauntlet with its experimental pneumococcal vaccine, VAX-31, going toe-to-toe with Pfizer’s Prevnar 20 in an early-stage trial. And let’s just say, the results are giving Pfizer’s brass plenty to worry about at their next earnings call.
VAX-31 didn’t just keep pace—it outperformed Prevnar 20, showing a stronger immune response against 18 out of 20 strains of those pesky streptococcus bacteria. This isn’t just a pat on the back; it’s a flashing neon sign that a market shake-up is on the horizon.
For those not yet familiar with Vaxcyte, let’s break it down. This biotech upstart is developing next-generation vaccines designed to do more than just fight infections—they’re engineered to outsmart them.
Vaxcyte’s secret weapon? That would be their XpressCF platform, a game-changing cell-free protein synthesis technology that’s rewriting the rules of vaccine development.
Unlike conventional methods that rely on live cells to produce proteins—a process that can be as slow and finicky as trying to bake a soufflé in a wind tunnel—XpressCF takes a completely different approach.
Essentially, XpressCF makes it possible to create complex proteins in a controlled environment without the need for live cells.
By extracting and using only the essential components from Escherichia coli bacteria, Vaxcyte can synthesize proteins with a level of precision that’s simply unattainable with traditional cell-based systems.
This not only speeds up the production process—cutting out the unpredictable nature of working with live cells—but also allows Vaxcyte to fine-tune the proteins to be more effective against the bacteria they’re targeting.
And as for the result? Vaccines that are not just produced faster, but are also more precisely engineered to take down the most stubborn pathogens. It’s like swapping out a sledgehammer for a scalpel—more targeted, more effective, and less room for error.
This leap forward in technology means Vaxcyte can potentially offer broader protection with fewer side effects, all while dodging the common production headaches that have plagued traditional vaccine development for decades.
And, as expected, Vaxcyte’s pipeline is packing some serious heat with VAX-31 and VAX-24, targeting 31 and 24 strains of IPD, respectively.
These vaccines are aimed squarely at protecting the most vulnerable—infants and adults over 50—from these dangerous infections.
While VAX-24 is leading the charge with Phase 2 trials already showing promising results, VAX-31 is closing in fast and could be the breakout star when it hits the market.
And here’s where it gets even more interesting. Vaxcyte has its sights set on a Phase 3 trial for VAX-31 in adults by mid-2025, with data expected to roll in by 2026.
If everything goes according to plan, Vaxcyte could have not one but two blockbuster vaccines ready to rock the market by 2027 — just as Pfizer and Merck (MRK) might be getting a little too comfortable in their respective corners.
Now, let’s pivot to the bigger picture. Pfizer’s Prevnar 20 and its predecessor, Prevnar 13, are nothing short of cash cows, raking in $6.4 billion in 2023—more than all but two other Pfizer products.
Merck isn’t just sitting on the sidelines, either. Their Vaxneuvance and Pneumovax 23 contributed a cool $1.1 billion to the bottom line last year.
With VAX-31 stepping into the ring, though, it’s like David just found a very large and very effective rock.
But before we get too far ahead of ourselves, let’s remember that biotech is a high-stakes game. The risks are real, and the path to FDA approval is littered with potential setbacks.
Still, Vaxcyte’s got a few things going for it that could make this a winning bet.
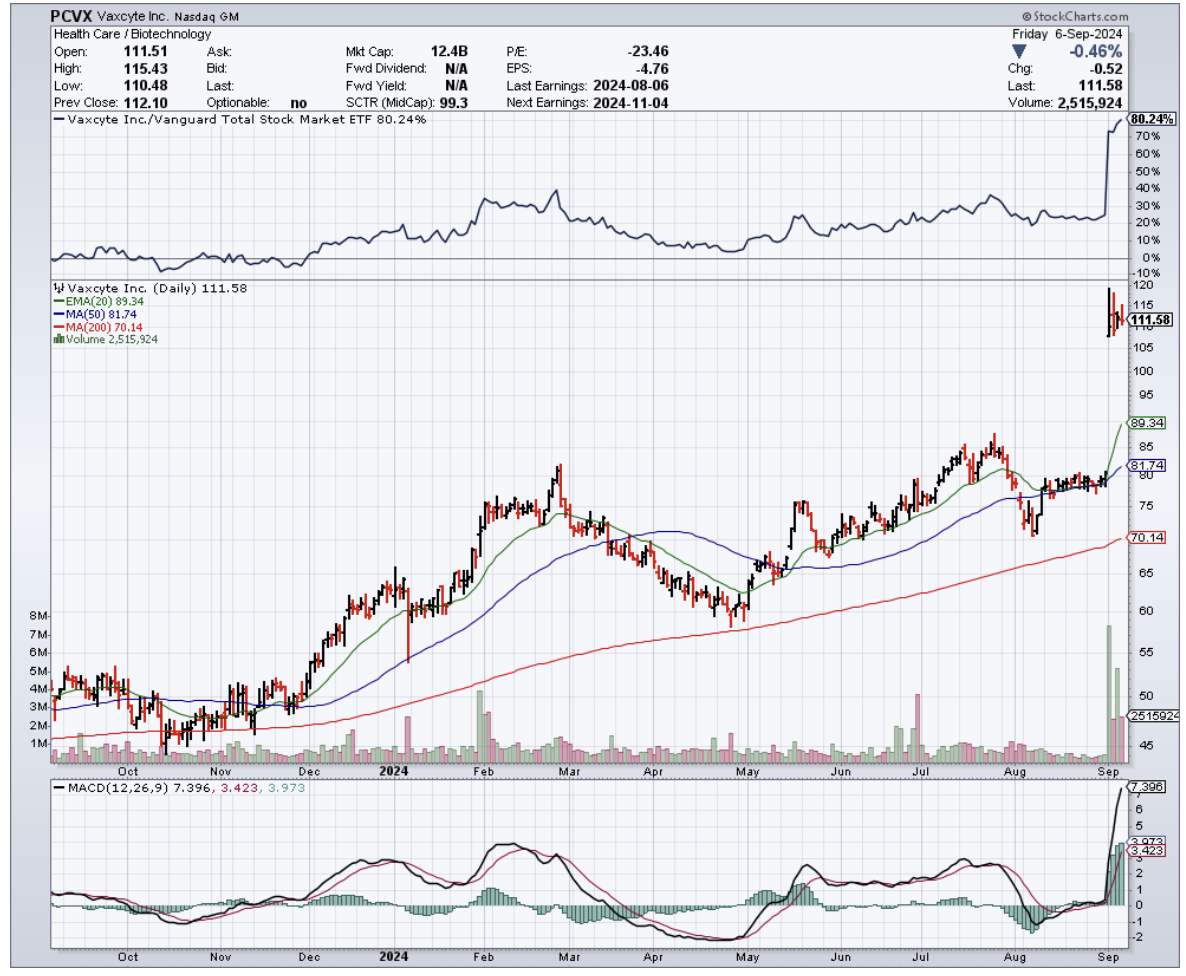
For starters, they’ve got $518.7 million in cash, $934 million in short-term investments, and zero debt. That’s a war chest of $1.5 billion to fund their march toward FDA approval without breaking a sweat.
From a valuation standpoint, Vaxcyte’s market cap is sitting pretty at $12.5 billion, and while some might balk at their price-to-book ratio of 6.3 compared to the sector’s median of 2.4, I’d argue that you get what you pay for.
After all, you’re not just buying into a company. If anything, you’re actually buying into a potential industry leader. And in a market where the reigning king is starting to sweat, that might be the most valuable investment of all.