Waiting for This Biotech to Stop Monkeying Around
Almost immediately after US President Joe Biden advised that “everybody” should be concerned over the new worldwide outbreak of the monkeypox virus, the shares of biotechnology and healthcare companies working on monkeypox treatments and vaccines started to rise.
Shares of Danish company Bavarian Nordic (BVNKF), the only monkeypox vaccine developer approved in the US, were up 5.8% in premarket following the announcement.
Bavarian Nordic’s vaccine, called Jynneos, uses a live version of the smallpox virus, which has been altered so that it no longer can replicate in the recipient’s body or cause any infection.
Instead, it has been engineered to activate the immune system and prepare the body’s defenses to fight off smallpox and monkeypox viruses.
Based on data from Africa, two shots of Jynneos, administered 28 days apart, recorded up to 85% in terms of efficacy against monkeypox.
In 2019, Jynneos received regulatory approval from the US FDA for both smallpox and monkeypox.
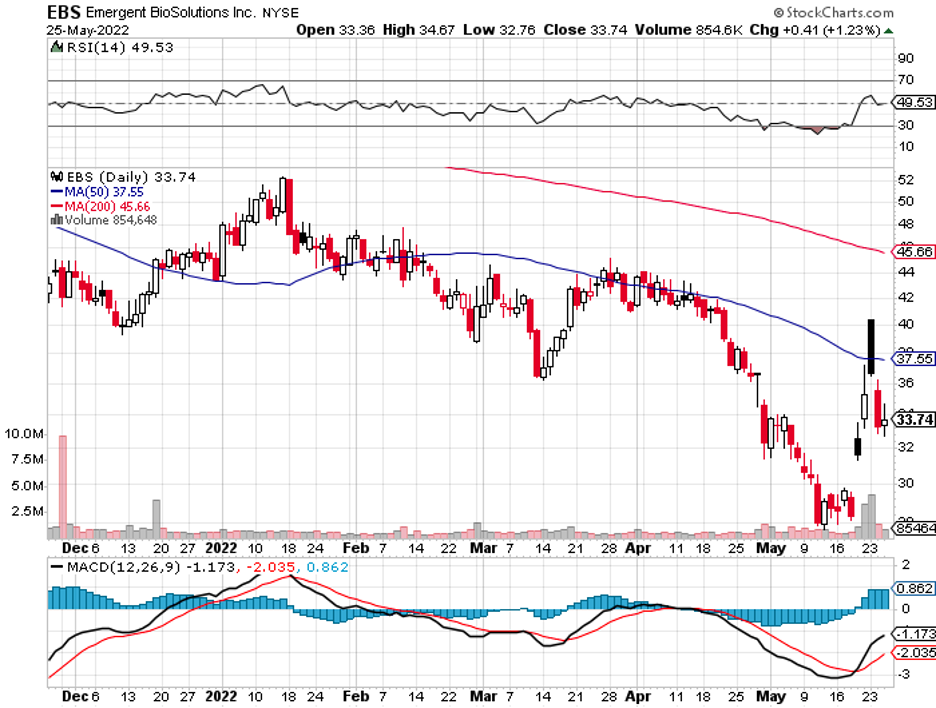
Aside from Bavarian Nordic, shares of Emergent BioSolutions (EBS) also rose by 11.8% following Biden’s announcement.
While Emergent has no vaccine specifically for monkeypox, it has a smallpox vaccine that can be used to prevent monkeypox.
It can be recalled that Emergent BioSolutions has been an exiled ticker after the US Congress launched an investigation on the manufacturing issues in its Bayview Facility in 2021.
Although the company has managed to clean up that mess and is back to working with Johnson & Johnson (JNJ) to produce COVID-19 vaccines, EBS has yet to return to investors’ good graces.
While the scale of the threat has yet to be determined, the US has secured contracts for Jynneos and Emergent BioSolutions’ vaccine and is already stockpiling in case of an outbreak.
What’s curious, though, is that another company has benefited from this announcement despite not having any monkeypox or even smallpox vaccine candidates.
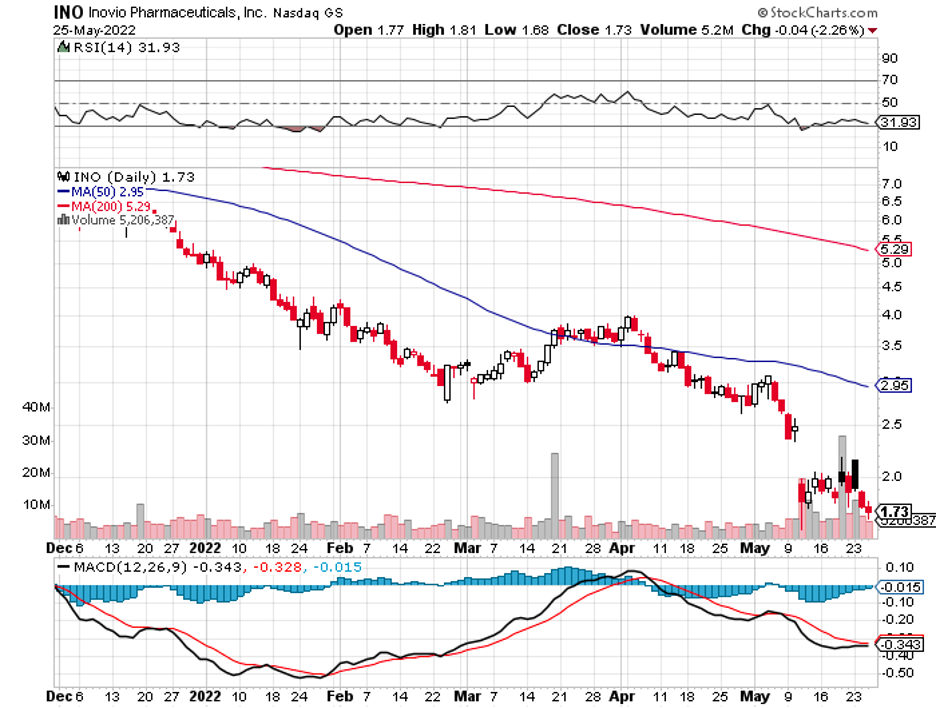
Inovio Pharmaceuticals (INO) shares rose by 12.2% following the announcement—a surge that couldn’t be adequately explained since the company has no relevant product and does not seem to have any program even remotely linked to this potential outbreak.
As far as I can tell, the last time Inovio even mentioned monkeypox was in 2010 when it discussed a potential experiment on a vaccine that could protect nonhuman primates against the virus. However, nothing came out of that plan either.
If Inovio sounds familiar to you, it’s probably because it was one of the frontrunners in the early days of the COVID-19 vaccine race.
However, it eventually lagged behind the likes of Moderna (MRNA), Pfizer (PFE), BioNTech (BNTX), and AstraZeneca (AZN).
One primary reason for this is the FDA’s decision to suspend Inovio’s Phase 3 trial in late 2020, with the study only resuming sometime in 2021.
As if that’s not enough, Inovio also faced some internal battles following the resignation of its CEO.
Now, the company has shifted gears and plans to offer its COVID-19 candidate as a booster shot instead of a primary vaccine.
The change of plans regarding the COVID-19 vaccine might be disappointing for some, but it’s essential to be realistic about expectations.
At the moment, the vaccine landscape has been dominated by Pfizer and Moderna, with AstraZeneca and Johnson & Johnson gaining ground as well.
Just recently, another challenger joined the fray: Novavax (NVAX).
Needless to say, the COVID-19 vaccine market is becoming crowded, and the competition is getting more intense.
Considering that Inovio has yet to catch up with the development of its candidate, it would be unwise to challenge the already established developers dominating the market today.
Hence, offering its COVID-19 candidate as a booster would provide it with higher marketability since health experts encourage people to mix and match their vaccines.
Outside these efforts, Inovio is a leader in developing DNA plasma-based vaccines. Before the pandemic, the company had been working on an extensive pipeline using this technology.
One of the most promising DNA-based vaccines from Inovio is VGX-3100, which targets an HPV-triggered disease called cervical dysplasia. Simply, this is a pre-cancer condition.
Inovio’s candidate is the first-ever DNA-based treatment that reached Phase 3 trials and reaped positive results.
This is an exciting development, especially in light of Inovio’s partnership with Qiagen (QGEN), as the two can leverage their work to determine which patients are at risk.
Basically, Inovio and Qiagen might just be on the verge of coming out with a preventive vaccine for cancer.
If things go according to plan, the data should be released by the second half of 2022. In terms of price, VGX-3100 is expected to cost roughly $10,000.
Aside from these, Inovio is also collaborating with Regeneron (REGN) to develop a cure for glioblastoma, an incredibly aggressive type of brain cancer. So far, Phase 2 trial results look promising, and the partners are on their way to progressing to Phase 3.
Inovio’s pipeline covers many DNA vaccines targeting infectious diseases and cancers. Most are still in the early phases of development.
While the programs in Phase 2 and 3 trials are promising, I think it’s still too early to predict whether Inovio is truly capable of delivering on its promises.
I know that Inovio shares look like such a bargain these days, especially if the company ends up receiving regulatory approvals in the coming months, but I’m not yet fully convinced.
Overall, Inovio is worth considering right now. It’s definitely on my list.
But before I commit, I’d like to see at least whether the company’s COVID-19 and HPV pipelines can move past the latest headwinds and advance to the next levels.