This is No Monkey Business
Hot on the trail of Moderna (MRNA), AstraZeneca (AZN), and Pfizer (PFE), biotechnology and healthcare titan Johnson & Johnson (JNJ) recently shared its progress on its COVID-19 vaccine candidate.
Recent reports show that JNJ’s vaccine protected the monkeys enrolled in its experiment from SARS-CoV-2 infection.
The success of this experiment pushed the company’s coronavirus vaccine efforts to start with human trials in the US and in Europe by the third quarter of 2020.
Hopefully, the JNJ will be able to provide conclusive data on its human trials by September.
Since the results of the human trials will take months before getting released, the efficacy and potency of JNJ’s vaccine can only be determined based on the available monkey data.
According to these, the JNJ vaccine might be more similar with Merck’s (MRK) candidate. That is, it might only require one injection compared to at least two doses required by its fellow vaccine makers.
Basically, this vaccine candidate involves a common cold virus called adenovirus or Ad26.
This virus is then modified to carry the coronavirus spike protein genetic material. When injected into humans, the modified Ad26 then slips into the cells and triggers the body to produce the coronavirus proteins.
Since Ad26 has been modified to only mimic the SARS-CoV-2, it cannot replicate but can trigger the body into putting up defenses against the COVID-19 virus.
The Ad26 vaccine uses the same technology the company applied in its Ebola vaccine sent to the Democratic Republic of Congo in 2019.
JNJ’s vaccine candidate received approval from European regulators in July, making it the first-ever virus-assisted gene delivery treatment approved for any disease.
Since March, JNJ has been working on at least seven variations of the Ad26 vaccine. The idea is to come up with various candidates that will target different types of patients.
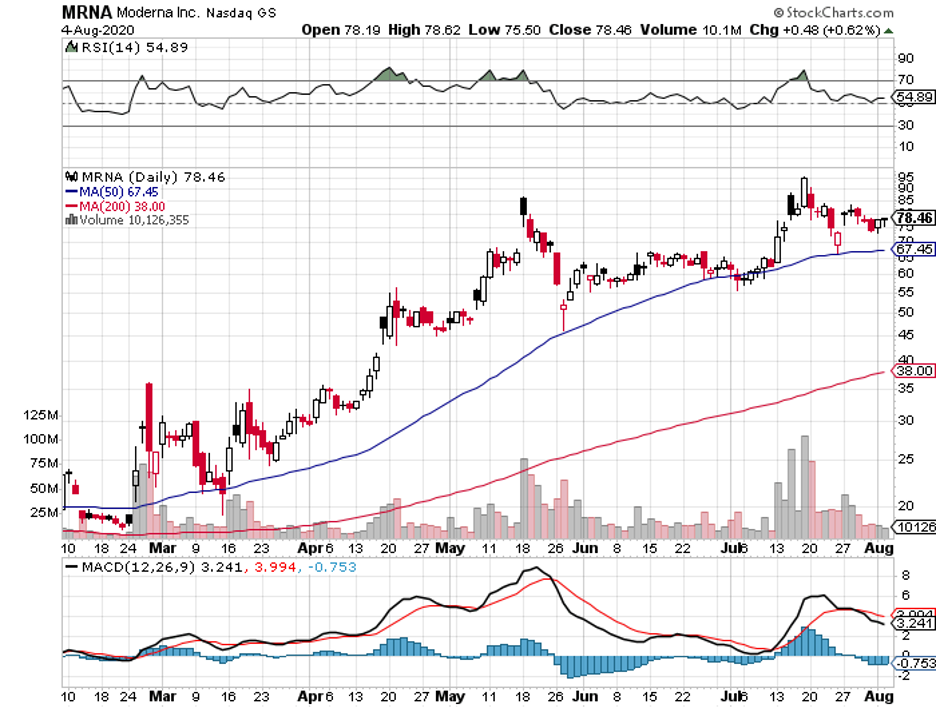
Aside from JNJ, COVID-19 vaccine leader Moderna has also released its monkey data for its vaccine trials.
In the Moderna experiment, the monkeys were given two shots of the vaccine in the course of four weeks. After a month, the same animals were infected with the SARS-CoV-2.
The results showed that no trace of the coronavirus was found in some of the vaccinated monkeys. While others still got infected, the virus gradually disappeared over time without any treatment.
Another COVID-19 frontrunner, Inovio Pharmaceuticals (INO), also released its monkey data.
The report covered four months from the day the monkeys were injected with Inovio’s vaccine candidate. According to the company’s findings, the infected animals only had traces of the coronavirus in their noses and lungs.
Other than JNJ, Moderna, and Inovio, both Pfizer and AstraZeneca also shared promising monkey data.
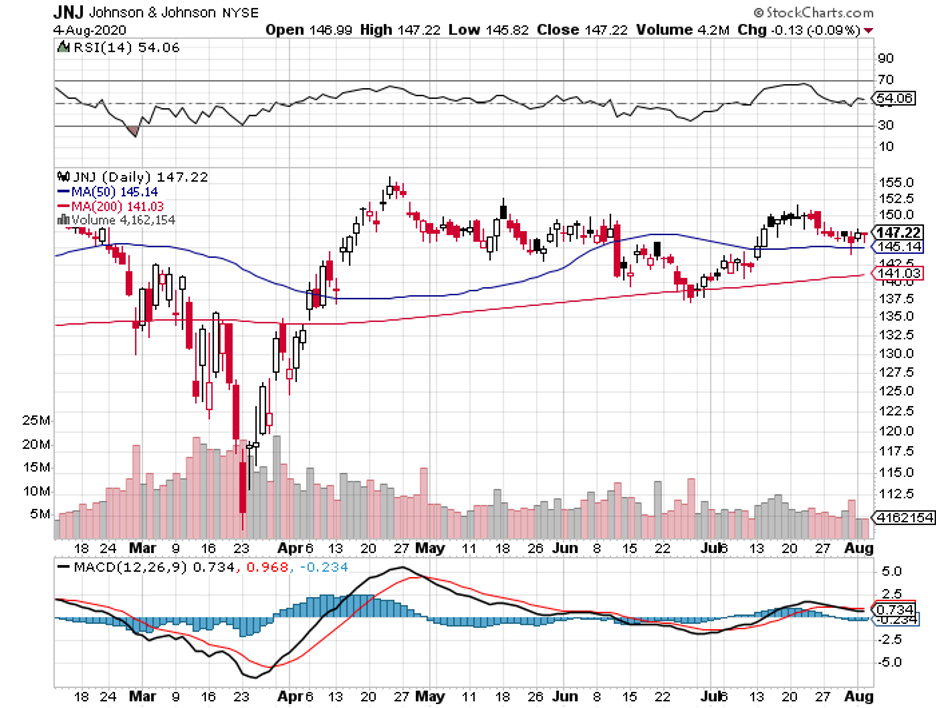
With a market capitalization of roughly $388 billion, JNJ is considered as one of the biggest biotechnology and healthcare companies in the world.
Apart from that, JNJ is one of the only two companies with an AAA credit rating. The other company is Microsoft (MSFT).
Needless to say, investing in an AAA-rated business with an impressive balance sheet makes buying JNJ stock an extremely low-risk and safe financial move.
JNJ has also been consistently regarded as a blue-chip dividend stock, offering a dependable 2.7% dividend.
While this may not be the highest yield you can get in the industry, JNJ has proven itself capable of handling the ups and downs of the market.
In fact, while most of the companies struggle to keep their heads above water, JNJ has surprisingly suffered minimal impact from this pandemic.
The company reported $20.7 billion in total revenue in the first quarter of 2020, showing off a 3.3% year-over-year climb in earnings and a 56.1% increase in earnings per share.
In JNJ’s second-quarter report, the company boosted its 2020 full-year guidance, with pharmaceutical revenue jumping to $18 billion and posting $3.6 billion in profits despite the 10.8% drop in sales.
One caveat when thinking about investing in JNJ is its widely reported talcum powder scandal. However, this issue poses no significant risk to the stock price since JNJ has already been penalized with billions of dollars.
More importantly, the company disclosed that it would no longer be selling baby powder. Clearly, the issue has been put to rest.
Regardless of how the COVID-19 vaccine race works out for JNJ, the company remains a solid investment. Its diverse product line alone makes it a financially resilient business. JNJ offers products from household items like Tylenol and Band-Aids to blockbuster drugs like severe psoriasis treatment Stelara.
Although JNJ is not particularly cheap at the moment, this stock is one of those investments that should be acquired at every pullback.
Since it is somewhat overvalued right now, my advice is to wait for a stock price correction soon and then pounce on the opportunity to own shares in this AAA-rated company.