With the flu season just around the corner and herd immunity nowhere in sight, the pressure to develop a COVID-19 vaccine becomes even more urgent. From where things stand right now though, it looks like we could have a vaccine either already available on the market or ready to hit the market around this time in 2021.
We know we’ll need hundreds of millions of vaccine doses, and the majority of the vaccine programs today are getting built on industrial-scale vaccine platforms. This is positive news.
On an even more positive update, a handful of biotechnology and health care companies are now on late-stage testing for the COVID-19 vaccine.
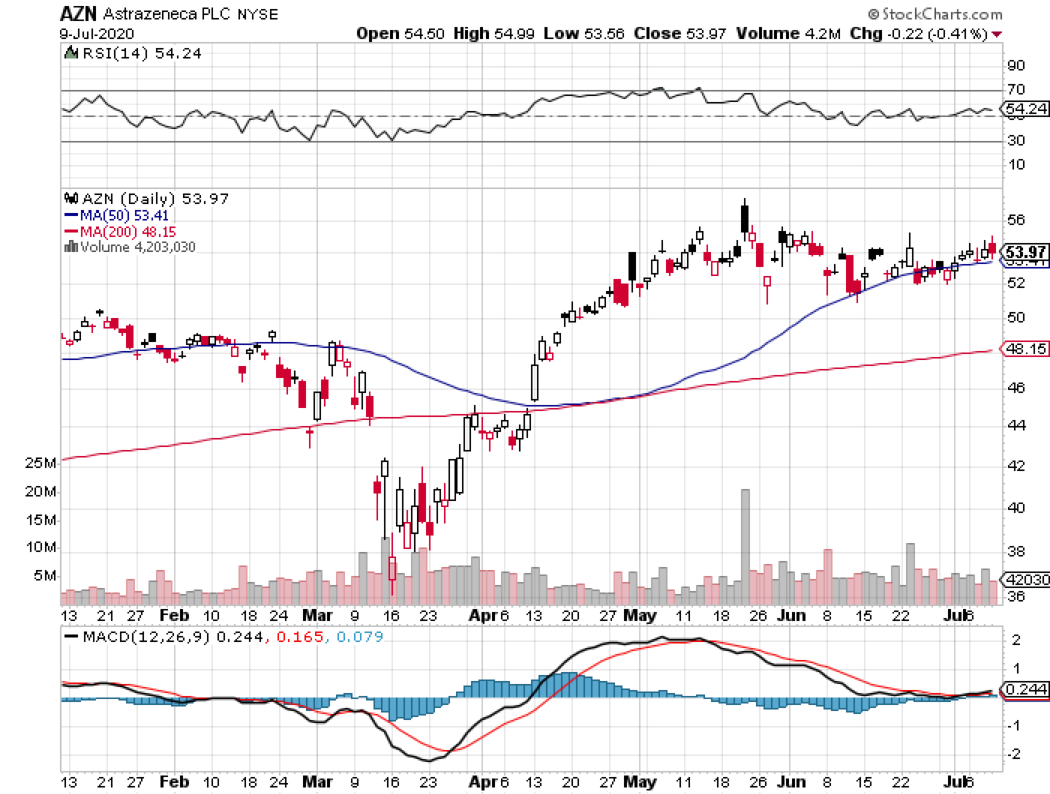
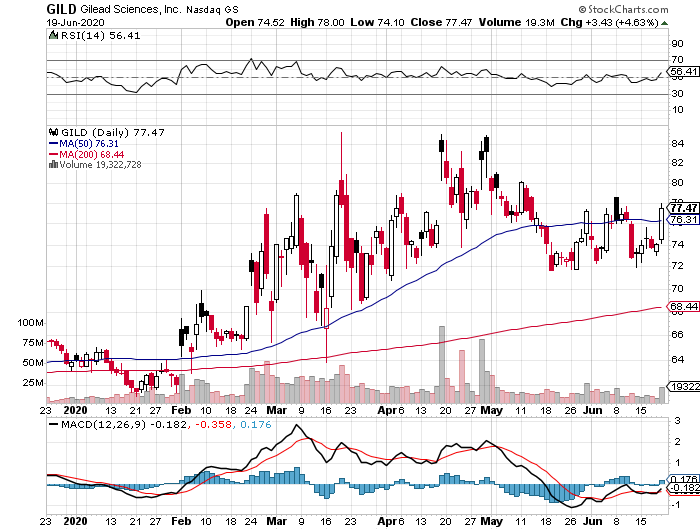
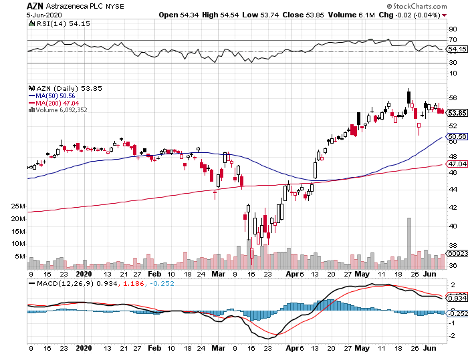
Leading the charge so far is AstraZeneca (AZN), which received $1.2 billion in financial assistance courtesy of the US government’s Operation Warp Speed program.
AstraZeneca is working on an experimental vaccine, called AZD1222, with the University of Oxford and China National Pharmaceutical Group (Sinopharm).
So far, this is the only COVID-19 vaccine candidate in late-stage Phase 3 trials.
The trials are scheduled to be conducted in different countries, with some already in progress in South Africa, Brazil, and of course, the UK.
The stage will enroll over 10,000 people in the UK alone. The goal is to determine AZD1222’s efficacy in a sizeable group aged 18 and older.
What we know about AstraZeneca’s vaccine candidate is that it’s created from a weakened version of adenovirus, which comes from one of the virus types that causes the common cold. It also includes genetic material from COVID-19, which was added to help the patient’s body recognize the pathogen and trigger a defense mechanism to fight off the infection.
Researchers say that the best-case scenario is for the Phase 3 efficacy results of the AstraZeneca vaccine to be available by this fall.
However, AstraZeneca remains an attractive stock even sans its Covid-19 program thanks to its remarkable drug pipeline. With the foresight to stockpile drugs during this pandemic, the company’s earnings are projected to continuously grow.
In the past five to six years, AstraZeneca has been aggressive in investing in its pipeline to combat patent losses. Now, the company joins Roche (RHHBY) and Eli Lilly (LLY) in the list of companies with the most innovative candidates that are poised to launch commercial products capable of driving growth in the next decade.
A notable growth driver for AstraZeneca is its cancer franchise, particularly its key drug Tagrisso, which is set to tap into a massive market.
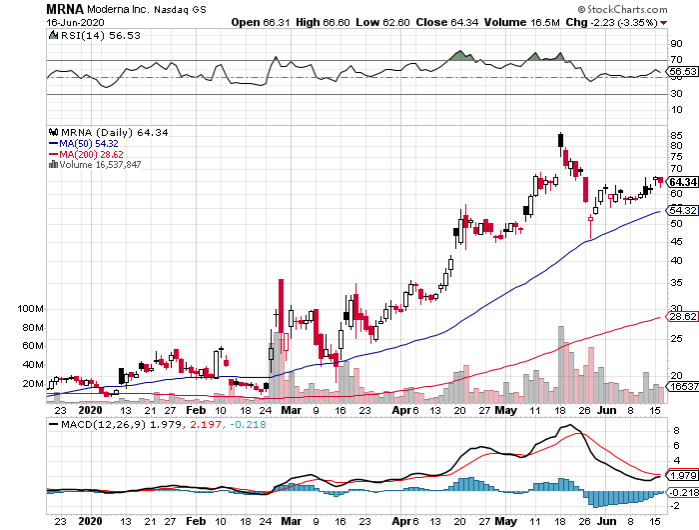
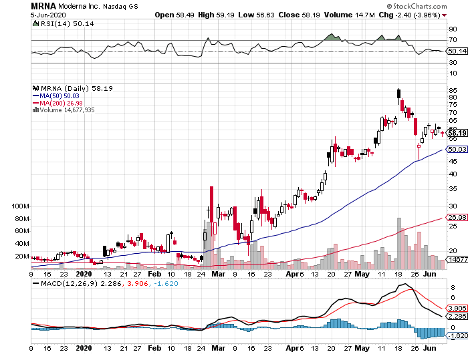
Before AstraZeneca was dubbed the leader in the COVID-19 vaccine race, there was Moderna (MRNA). Actually, this small biotechnology company is also expected to begin its late-stage Phase 3 trial in July.
Like AstraZeneca, Moderna is also one of the companies included in the Operation Warp Speed project and received $483 million from the government.
Unlike AstraZeneca, Moderna appears to be experiencing delays due to conflicts between the company’s experts and the US government scientists.
While Moderna shares jumped by over 200% since the pandemic started, these reported tensions represent a risk for its investors. It is particularly alarming because the company is a clinical-stage biotechnology company with no marketed products.
Although Moderna’s timeline remains to be the most aggressive, it could easily drown in the competition.
Keep in mind that other companies competing for the top spot in the COVID-19 race are all established and armed with extensive experience in launching new drugs to market. The list includes Pfizer (PFE), which has a market capitalization of $185.86 billion, and Johnson & Johnson (JNJ) with $375.40 billion.
Needless to say, the inexperience of companies like Moderna could prove to be a handicap in this highly competitive race.